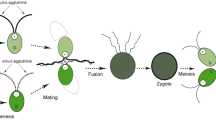
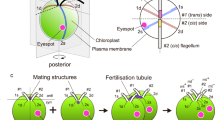
Abstract
Routinely prepared gametes (by flooding 3 week-old agar cultures) showed about 80% mating competence if the opposite sexual partners were mixed together. The mating competence exhibited a strict dependence on the composition of the solution in which the cells were suspended before mixing; it decreased progressively with increasing concentration of nitrates. In contrast, no inhibiting effect was found if urea was used as the source of nitrogen. Other ions present in nutrient media did not show any effect. Mating activity varied according to the spectral composition of light, being higher with a blue light than with a red one. Blue light caused accumulation ofvis-à-vis pairs, which were blocked to form zygotes. Freshly released daughter cells in vegetatively grown synchronous cultures had a dual nature—vegetative and sexual one. In these daughter cells, similar rules were found for governing of mating competence to those valid for standard gametes obtained from flooded agar cultures. High mating competence was found in daughter cells released the during dark period in distilled water, nitrate-free media, in the presence of Mg2+ or Ca2+ ions, or in media containing urea. The conditions during which daughter cells are released and the conditions under which they mate can be considered crucial for expression of gametic nature as a mating competence.
Similar content being viewed by others
References
Beck C.F., Acker A.: Gametic differentiation ofChlamydomonas reinhardtii.Plant Physiol. 98, 822–826 (1992).
Beck C.F., Haring M.A.: Gametic differentiation ofChlamydomonas.Internat. Rev. Cytol. 168, 259–301 (1996).
Kates J.R., Jones R.F.: The control of gametic differentiation in liquid cultures ofChlamydomonas.J. Cell. Comp. Physiol. 63, 157–164 (1964).
Kooijman R., de Wildt P., Homan W.L., Musgrave A., van den Ende H.: Light effects flagellar agglutinability inChlamydomonas eugametos by modification of the agglutinin molecules.Plant Physiol. 86, 216–223 (1988).
Matsuda Y., Shimada T., Sakamoto Y.: Ammonium ions control gametic differentiation and dedifferentiation inChlamydomonas reinhardtii.Plant Cell Physiol. 9, 1–6 (1992).
Molendijk A.J., van Egmond P., Haring M.A., Klis F.M., van den Ende H.: Characterization of the cell cycle in synchronous cultures ofChlamydomonas eugametos in relation to gametogenesis.J. Gen. Microbiol. 138, 1941–1947 (1992).
Sager R., Granick S.: Nutritional control of sexuality inChlamydomonas reinhardtii.J. Gen. Physiol. 37, 729–742 (1954).
Schuring F., Brederoo J., Musgrave A., van den Enge H.: Increase in calcium triggers mating structure activation inChlamydomonas eugametos.FEMS Microbiol Lett. 71, 237–240 (1990).
Schuring F., Musgrave A., Elders M.C.C., Teunissen Y., Homan W.L., van den Ende H.: Fusion-defective mutants ofChlamydomonas eugametos.Protoplasma 162, 108–119 (1991)
Weissig H., Beck C.F.: Action spectrum for the light-dependent step in gametic differentiation ofChlamydomonas reinhardtii.Plant Physiol. 97, 118–121 (1991).
Zachleder V., Jakobs M., van den Ende H.: Relationship between gametic differentiation and the cell cycle in the green algaChlamydomonas eugametos.J. Gen. Microbiol. 137, 1333–1339 (1991).
Zachleder V., Šetlík I.: Effect of irradiance on the course of RNA synthesis in the cell cycle ofScenedesmus quadricauda.Biol. Plant. 24, 341–353 (1982).
Zachleder V., van den Ende H.: Cell cycle events in the green algaChlamydomonas eugametos and their control by environmental factors.J. Cell Sci. 102, 469–474 (1992).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zachleder, V., Hendrychová, J., Bišová, K. et al. Factors affecting the mating competence in the unicellular green algaChlamydomonas eugametos (Volvocales) . Folia Microbiol 47, 69–72 (2002). https://doi.org/10.1007/BF02818568
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02818568