Abstract
It is well known that cellulolytic enzymes hardly attack lignocellulose. Hitherto knowledge, however, has not yet fully elucidated the mechanisms of the inhibition; whether it depends on a steric hindrance or protein-binding inhibition. For the purpose of acceleration of enzymatic digestion of lignocellulose biomass, the study was carried out using partially purified cellulase of Meicelase and milled wood lignin from soft or hard wood.
Milled wood lignin was prepared from soft wood,Picea jezoensis, or from hard wood,Quercus serrata, by the method of Björkman. Lignin was dissolved in methyl cellosolve, since the solvent has little effect on cellulolytic activities, and enzymic activities were followed with partially purified Meicelase.
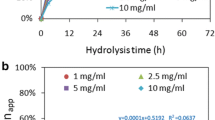
The results show that cellobiose activities of enzymes fromTrichoderma viride are markedly depressed by both lignin of soft and hard wood, and the former lignin inhibits the reaction stronger than the latter. Lignins show no inhibition in the reaction with CMC and debris of filter paper.
Liberation of glucose from filter paper by the enzyme in the presence of lignin is also depressed, and addition of cellobiase instead of lignin in the above enzyme reaction shows the delay of time for breakdown of filter paper. This phenomenon indicates that lignin inhibits enzymic decomposition of cellobiose in the degradation pathway of cellulose and accumulated cellobiose gives a feedback function to digestion of fibers of paper.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hidaka, H., Takizawa, T., Fujikawa, H. et al. A study on the inhibition of cellulolytic activities by lignin. Appl Biochem Biotechnol 9, 367 (1984). https://doi.org/10.1007/BF02798979
Issue Date:
DOI: https://doi.org/10.1007/BF02798979