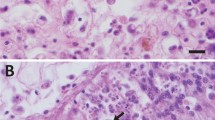
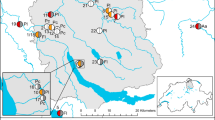
Abstract
Delaware's Inland Bays comprise a large estuarine system with a restricted access to the Atlantic Ocean (Indian River Inlet). As part of a local oyster stock enhancement and restoration effort, we conducted a survey for the protozoan pathogenPerkinsus marinus (Dermo) in oysters from a newly established reef. Using standardized methods for the polymerase chain reaction (PCR) amplification of the non-transcribed spacer (NTS) region, we were surprised to find no detectable titers of this pathogen in the 30 oysters sampled in the first year of the project. The detection threshold of the PCR coupled with chemiluminescent detection was 30 fgP. marinus NTS DNA. We were able to detect a trace presence of this pathogen in a few hard clams (Mercenaria mercenaria) from the same locale, indicating that aPerkinsus sp. was present in the Inland Bay system. Subsequent monitoring of the reef system using a fluid thioglycollate assay over 3 yr revealed no epizootic outbreaks of this pathogen within the planted oyster population. Two large mortality episodes that did appear in the oyster population were attributable to abiotic conditions and not pathogen exposure. This study emphasizes that all potential sources of mortality in the environment are important to consider when designing oyster seeding projects. In the Delaware Inland Bays,P. marinus does not appear to have a large enough oyster host population to become a significant disease threat at present. Because of the low parasite incidence levels in the Inland Bay system in 2000, the James Farm oyster reef restoration project presents an ideal model system to follow the population dynamics between an oyster-host population and a latent or reservoir pathogen population.
Similar content being viewed by others
Literature Cited
Andrews, J. D. 1996. History ofPerkinsus marinus, a pathogen of oysters in Chesapeake Bay 1950–1984.Journal of Shellfish Research 15:13–16.
Audemard, C., K. S. Reece, andE. M. Burreson. 2004. Real-time PCR for detection and quantification of the protistan parasitePerkinsus marinus in environmental waters.Applied and Environmental Microbiology 70:6611–6618.
Azevedo, C. 1989. Fine structure ofPerkinsus atlanticus n. sp. (Apicomplexa, Perkinsea) parasite of the clamRuditapes decussatus from Portugal.Journal of Parasitology 75:627–635.
Blackbourn, J., S. M. Bower, andG. R. Meyer. 1998.Perkinsus qugwadi sp. Nov. (incertae sedis), a pathogenic protozoan parasite of Japanese scallops,Patinopecten yessoensis, cultured in British Columbia, Canada.Canadian Journal of Zoology 76:942–953.
Burreson, E. M., K. S. Reece, andC. F. Dungan. 2005. Molecular, morphological, and experimental, evidence support the synonymy ofPerkinsus chesapeaki andPerkinsus andrewsi.Journal of Eukaryotic Microbiology 52:258–270.
Calvo, L. M. R., G. W. Calvo, andE. M. Burreson. 2003. Dual disease resistance in a selectively bred eastern oyster,Crassostrea virginica, strain tested in Chesapeake Bay.Aquaculture 220:69–87.
Calvo, L. M. R., R. L. Wetzel, andE. M. Burreson. 2001. Development and verification of a model for the population dynamics of the protistan parasite,Perkinsus marinus, within its host, the eastern oyster,Crassostrea virginica, in Chesapeake Bay.Journal of Shellfish Research 20:231–241.
Casas, S. M., A. Grau, K. S. Reece, K. Apakupakui, C. Azevedo, andA. Villalba. 2004.Perkinsus mediterraneus n. sp. a protistan parasite of the European flat oysterOstrea edulis (L.) from the Coast of Balearic Islands, Mediterranean Sea.Diseases of Aquatic Organisms 58:231–244.
Casas, S. M., A. Villalba, andK. S. Reece. 2002. Study of perkinsosis in the carpet shell clamTapes decussatus in Galicia (NW Spain). I. Identification of the aetiological agent and in vitro modulation of zoosporulation by temperature and salinity.Diseases of Aquatic Organisms 50:51–65.
Coss, C. A., J. A. F. Robledo, G. M. Ruiz, andG. R. Vasta. 2001. Description ofPerkinsus andrewsi n. sp isolated from the baltic clam (Macoma balthica) by characterization of the ribosomal RNA locus, and development of a species-specific PCR-based diagnostic assay.Journal of Eukaryotic Microbiology 48:52–61.
Dungan, C. F., R. M. Hamilton, K. L. Hudson, C. B. McCullough, andK. S. Reece. 2002. Two epizootic diseases in Chesapeake Bay commercial clamsMya arenaria andTagelus plebeius.Diseases of Aquatic Organisms 50:67–78.
Goggin, C. L.. 1994. Variation in the 2 internal transcribed spacers and 5.8S ribosomal-RNA from 5 isolates of the marine parasitePerkinsus (Protista, Apicomplexa),Molecular and Biochemical Parasitology 65:179–182.
Harvell, C. D., K. Kim, J. M. Burkholder, R. R. Colwell, P. R. Epstein, D. J. Grimes, E. E. Hoffmann, E. K. Lipp, A. D. M. E. Osterhaus, R. M. Overstreet, J. W. Porter, G. W. Smith, andG. R. Vasta. 1999. Review: Marine ecology-Emerging marine diseases-Climate links and anthropogenic factors.Science 285: 1505–1510.
Hinegardner, R.. 1974. Cellular DNA content of the Mollusca. 47A:447–460.Comparative Biochemistry and Physiology A 47A:447–460.
Hofmann, E. E., E. N. Powell, J. M. Klinck, andG. Saunders. 1995. Modeling diseased oyster populations. I. ModelingPerkinsus marinus infections in oysters.Journal of Shellfish Research 14:121–151.
La Peyre, M. K., A. D. Nickens, A. K. Volety, G. S. Tolley, andJ. F. La Peyre. 2003. Environmental significance of freshets in reducingPerkinsus marinus infection in eastern oystersCrassostrea virginica: Potential management applications.Marine Ecology Progress Series 248:165–176.
Lester, R. J. D. andG. H. G. Davis. 1981. A newPerkinsus species (Apicomplexa, Perkinsea) from the abaloneHaliotis ruber.Journal of Invertebrate Pathology 37:181–187.
Mackin, J. G., H. M. Owen, andA. Collier. 1950. Preliminary note on the occurrence of a new protistan parasite,Dermocystidium marinum n. sp. inCrassostrea, virginica (Gmelin).Science 111:328–329.
Marsh, A. G., J. D. Gauthier, andG. R. Vasta. 1995. A semiquantitative PCR assay for assessingPerkinsus marinus infections in the eastern oyster,Crassostrea virginica.Journal of Parasitology 81:577–583.
McLaughlin, S. M., E. E. Elsayed, andM. Faisal. 2000b. Analysis of extracellular proteins of twoPerkinsus sp. isolated from the softshell clamMya arenaria in vitro.Comparative Biochemistry and Physiology B—Biochemistry and Molecular Biology 126:587–598.
McLaughlin, S. M., B. D. Tall, A. Shaheen, E. E. Elsayed, andM. Faisal. 2000a. Zoosporulation of a newPerkinsus species isolated from the gills of the softshell clamMya arenaria.Parasite—Journal de la Societe Francaise Parasitologie 7:115–122.
Milbury, C. A., D. W. Meritt, R. I. E. Newell, andP. M. Gaffney. 2004. Mitochondrial DNA markers allow monitoring of oyster stock enhancement in the Chesapeake Bay.Marine Biology 145:351–359.
Murrell, A., S. N. Kleeman, S. C. Barker, andR. J. G. Lester. 2002. Synonymy ofPerkinsus olseni Lester and Davis, 1981 andPerkinsus atlanticus Azevedo, 1989 and an update on the phylogenetic position of the genusPerkinsus.Bulletin of the European Association of Fish Pathologist 22:258–265.
Perkins, F. O. 1993. Infectious diseases of molluscs, p. 255–288.In J. A. Couch (ed.), Pathobiology of Marine and Estuarine Organisms, Volume 1. CRC Press, Boca Raton, Florida.
Porter, C. H., andF. H. Collins. 1991. Species-diagnostic differences in a ribosomal DNA internal transcribed spacer from the sibling speciesAnopheles freeborni andAnopheles hermsi (Diptera: Culicidae).American Journal of Tropical Medicine and Hygine 45:271–279.
Powell, E. N., J. M. Klinck, andE. E. Hofmann. 1996. Modelling diseased oyster populations. 2. Triggering mechanisms forPerkinsus marinus epizootics.Journal of Shellfish Research 15:141–165.
Ray, S. M. 1996. Historical perspective onPerkinsus marinus disease of oysters in the Gulf of Mexico.Journal of Shellfish Research 15:9–11.
Reece, K. S., D. Bushek, K. L. Hudson, andJ. E. Graves. 2001. Geographic distribution ofPerkinsus marinus genetic strains along the Atlantic and Gulf coasts of the USA.Marine Biology 139:1047–1055.
Robledo, J. A. F., C. A. Coss, andG. R. Vasta. 2000. Characterization of the ribosomal RNA locus ofPerkinsus atlanticus and development of a polymerase chain reaction-based diagnostic assay.Journal of Parasitology 86:972–978
Robledo, J. A. F., J. D. Gauthier, C. A. Coss, A. C. Wright, andG. R. Vasta. 1998. Species-specificity and sensitivity of a PCR-based assay forPerkinsus marinus in the eastern oyster,Crassotrea virginica: A comparison with the fluid thioglycollate assay.Journal of Parasitology 84:1237–1244.
Robledo, J. A. F., A. C. Wright, A. G. Marsh, andG. R. Vasta. 1999. Nucleotide sequence variability in the nontranscribed spacer of the rRNA locus in the oyster parasitePerkinsus marinus.Journal of Parasitology 85:650–656.
Sambrook, J., E. F. Fritsch, andT. Maniatis (eds.). 1989. Molecular Cloning: A Laboratory Manual, 2nd edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Vasta, G. R., J. D. Gauthier, andA. G. Marsh. 1995. Molecular and biochemical adaptations of the protozoan pathogenPerkinsus marinus for its host,Crassostrea virginica: Current research and future perspectives (or “the best defense is a good offense”).Journal of Marine Biotechnology 3:35–41.
Villaiba, A., K. S. Reece, M. C. Ordás, S. M. Casas, andA. Figueras. 2004. Perkinsosis in mollusks: A review.Aquatic Living Resources 17:411–432.
Volety, A. K. andW. S. Fisher. 2000. In vitro killing ofPerkinsus marinus by hemocytes of oystersCrassostrea virginica.Journal of Shellfish Research 19:827–834.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kendall, L.R., Ewart, J.W., Ulrich, P.N. et al. Low incidence and limited effect of the oyster pathogen dermo (Perkinsus marinus) on an artificial reef in Delaware's Inland Bays. Estuaries and Coasts: J ERF 30, 154–162 (2007). https://doi.org/10.1007/BF02782975
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02782975