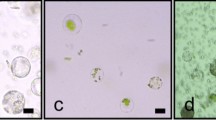
Abstract
Establishment of protoplast techniques for conifers is of crucial importance to forest biotechnology. Protoplasts can be used to regenerate whole trees and to study transcriptional regulation in woody plants by promoter analysis. Here we describe a technique for isolating protoplasts fromPinus pinaster shoots. Protoplast viability was determined by fluorescein diacetate staining. Transient gene expression was studied after electroporation of protoplasts with plasmid DNA containing a β-glucuronidase (GUS) reporter gene. The 35S promoter and a pine (cytosolic glutamine synthetase) promoter both were able to driveGUS expression, indicating applicability of conifer protoplasts as a model to study gene promoters using transient expression.
Similar content being viewed by others
References
Avila C, Muñoz-Chéapuli R, Plomion C, Frigerio J-M, and Céanovas FM (2000) genes encoding distinct cytosolic glutamine synthetases are closely linked in the pine genome. FEBS Lett 477: 237–243.
Avila C, Suéarez M-F, Géomez-Maldonado J, and Céanovas FM (2001) Spatial and t expression of two cytosolic glutamine synthetase genes in Scots pine: functional implications on nitrogen metabolism during early stages of conifer development. Plant J 25: 93–102.
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254
Céanovas FM, Cantéon FR, Gallardo F, Garcéia-Gutiéerrez A, and de V (1991) Accumulation of glutamine synthetase during early development of maritime pine (Pinus pinaster) seedlings. Planta 185: 372–378.
David H, David A, and Mateille T (1982) Evaluation of parameters affecting the yield, viability and division ofPinus pinater protoplasts. Physiol Plant 56: 108–113.
Duhoux E (1980) Protoplast isolation of gymnosperm pollen. Z Pflanzenphysiol 99: 207–214.
Faye E and David A (1983) Isolation and culture of gymnosperm root protoplasts (Pinus pinaster). Physiol Plant 59: 359–362.
Gupta PK and Durzan DJ (1986) Isolation and cell regeneration of protoplasts from sugar pine (Pinus lambertiana). Plant Cell Rep 5: 346–348.
Hakman IC, von Harnold S, and Fellner-Feldegg H (1986) Isolation and DNA analysis of protoplasts from developing female gametophytes ofPicea abies (Norway spruce). Plant Sci 38: 53–59.
Jefferson RA, Kavanagh TA, and Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile fusion marker in higher plants. EMBO J 7: 1265–1271.
Mitsuhara I, Ugaki M, Hirochika H, Oshima M, Murakami T, Gotoh Y, Katayose Y, Nakamura S, Honkura R, Nishimiya S, Ueno K, Mochizuki A, Tanimoto H, Tsugawa H, Otsuki Y, and Ohashi Y (1996) Efficient promoter cassettes for enhanced expression of foreign genes in dicotyledonous and monocotyledonous plants. Plant Cell Physiol 37: 49–59.
Patel KR, Shekhawat NS, Berlyn GP, and Thorpe TA (1984) Isolation and culture of protoplasts from cotyledons ofPinus coulteri. Plant Cell Tissue Organ Cult 3: 85–90.
Rotman BR and Papermaster RD (1996) Membrane properties of living cells as studied by enzymatic hydrolysis of fluorogenic esters. Proc Natl Acad Sci USA 55: 134–141.
Shen WJ, Williamson MS, and Forde BG (1992) Functional analysis of the promoter region of a nodule-enhanced glutamine synthetase gene fromPhaseolus vulgaris L. Plant Mol Biol 19: 837–846.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Géomez-Maldonado, J., Crespillo, R., éAvila, C. et al. Efficient preparation of maritime pine (Pinus pinaster) protoplasts suitable for transgene expression analysis. Plant Mol Biol Rep 19, 361–366 (2001). https://doi.org/10.1007/BF02772834
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02772834