Abstract
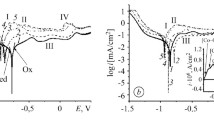
The corrosion resistance of some metal-like compounds in H2SO4 of concentration 9.7 M is studied. It is established that the composition TaC is the most corrosion-resistant. The method of stationary polarization curves is used to study the reaction of electric oxidation of hydrogen on sintered and thin-film TaC electrodes in H2SO4. It is shown that under the given conditions TaC is a catalyst of the reaction of oxidation of hydrogen.
Similar content being viewed by others
References
M. R. Tarasevich and G. V. Zhutaeva, “Electrocatalysis by refractory compounds,”Itogi Nauki Tekh., Ser. élektrokhimiya,22, 140–204 (1985).
G. V. Samsonov, V. K. Vitryanyuk, and F. I. Chaplygin,Tungsten Carbides [in Russian], Naukova Dumka, Kiev (1974).
G. V. Samsonov (ed.),Analysis of Refractory Compounds [in Russian], Gos. Nauch. Tekh. Izd. Chern. Tsvet. Met., Moscow (1962).
G. V. Samsonov and V. I. Konstantinov,Tantalum and Niobium [in Russian], Gos. Nauch. Tekh. Izd. Chern. Tsvet. Met., Moscow (1959).
O. I. Popova and G. T. Kabannik, “Acid resistance and methods for analyzing titanium, zirconium, niobium, and tantalum nitrides,”Zh. Neorg. Chem.,5(4), 930–934 (1960).
G. V. Samsonov and I. M. Vinnitskii,Refractory Compounds, A Reference Book, 2nd Edition [in Russian], Metallurgiya, Moscow (1976).
G. V. Samsonov,Nitrides [in Russian], Naukova Dumka, Kiev (1969).
R. D. Armstrong, A. F. Douglas, and D. E. Keene, “The anodic oxidation of hydrogen in aqueous acids by conducting compounds of the transition elements,”J. Electrochem. Soc.,118(4), 568–571 (1971).
K. Vetter,Electrochemical Kinetics [Russian translation], Khimiya, Moscow (1967).
Author information
Authors and Affiliations
Additional information
Translated from Ogneupory i Tekhnicheskaya Keramika, Nos. 1–2, pp. 11–14, January–February, 1999.
The first article of the series was published in No. 11 of 1998.
Rights and permissions
About this article
Cite this article
Lukashenko, T.A., Tikhonov, K.I. & Sher, é.M. Corrosion resistance and electrocatalytic activity of some refractory compounds of transition metals. Refract Ind Ceram 40, 10–13 (1999). https://doi.org/10.1007/BF02762436
Issue Date:
DOI: https://doi.org/10.1007/BF02762436