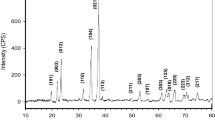
Abstract
The authors examine the boric oxide—ammonia route with special stress on the yield and composition of the intermediate addition compound (BN) x (B2O3) y (NH3) z . It has been concluded that B2O3 and NH3 present in the addition compound formed between 350°C and 900°C cannot be further reacted to convert the B2O3 into BN and the BN yield remains at around 66%. A formula (BN)12·7(B2O3)7·5NH3 has been suggested for the addition compound.
Similar content being viewed by others
References
Economy J, Anderson R V and Matcovich V I 1969Appl. Polym. Symp. 9 347
Ingles T A and Popper P 1960Special Ceramics (London: Heywood) p. 144
Mitra B and Sinha B C 1987Trans. Indian Ceram. Soc. 46 132
Mukerji J, Biswas S K and Dhargupta K K 1978Indian J. Technol. 16 156
Parker A and Healy C 1970Analyst 91 204
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Basu, A.K., Mukerji, J. Synthesis of boron nitride. Bull. Mater. Sci. 13, 165–171 (1990). https://doi.org/10.1007/BF02744942
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02744942