Abstract
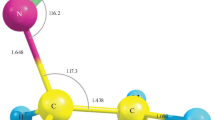
The density functional method (exchange correlation functional Becke3LYP) is used to calculate the potential energy profiles for the reaction of ozone with ethylene. It is shown that “direct” epoxidation of the C = C double bond demands high activation energy and is unlikely for both themwchemical and photochemical reactions of ozonolysis.
Similar content being viewed by others
References
S. D. Razumovskii and G. E. Zaikov,Ozone and Its Reactions with Organic Compounds [in Russian], Nauka, Moscow (1974).
G. Marcelin and P. R. Brooks,J. Am. Client. Soc,97, 1710–1715 (1975).
R. Atkinson, B. J. Finlayson, and J. N. Pitts,ibid. 95, 7592–7599 (1973).
B. J. Finlayson, J. N. Pitts, and R. Atkinson,ibid.,96, 5356–5367 (1974).
C. W. Gillies, J. Z. Gillies, R. D. Suenram, et al.,ibid.,113, 2412–2421 (1991).
S. Jonnalagadda, S. Chan, J. Carrido, et al.,ibid.,117, 562–563 (1995).
Y. Nakano, S. Saito, and Y. Morino.Bull. Chem. Soc. Jpn.,43 368–371 (1970).
V. M. Rayon and J. A. Sordo,Theor. Chem. Acc,99, 68–70 (1998).
H. M. Rohmer and B. Roos,J. Am. Chem. Soc,97, 2025–2030 (1975).
N. F. Tyupalo, V. Beloborodov, and Yu. B. Vysotskii,Dokl. Akad. Nauk SSSR,269. 377–380 (1983).
N. F. Tyupalo and Yu. B. Vysotskii,Zh. Org. Khim.,23, 1477–1480 (1987).
N. F. Tyupalo,Abstracts of Papers from the 2nd All-Union Symposium on Organic Synthesis. Moscow (1976), pp. 74–75.
N. F. Tyupalo and N. V. Bernashevskii.Dokl. Akad. Nauk SSSR,253. 896–898 (1980).
N. F. Tyupalo and A. F. Dmitruk,Dokl. Akad. Nauk USSR, Sei: B, No. 11, 51–55 (1986).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al.,GAUSSIAN-92IDFT, Revision G.2, Gaussian Inc., Pittsburgh (1993).
T. H. Dunning,J. Chem. Phys.,53. 2823–2840 (1970).
C. Möller and M. S. Plesset,Phys. Rev.,46, 618–635 (1934).
I. I. Zakharov, O. I. Kolbasina, T. N. Semenyuk. et al.,Zh. Stivkt. Khim.. 31, No. 2. 28–32 (1992).
I. I. Zakharov, O. I. Kolbasina. T. N. Semenyuk, et al.,ibid. ,34, No. 2, 26–30 (1992).
E. K. U. Gross and R. M. Drezier (eds.).Density Functional Theory, NATO ASI Series, Ser: B: Physics, Vol. 337, Plenum, New York (1995).
A. D. Becke.J. Chem. Phys.,98. 5648–5662 (1993).
C. Lee, W. Yang, and R. G. Parr,Phys. Rev. B. 37, 785–797 (1988).
K. S. Krasnov, N. V. Filippenko, V. A. Bobkova. et al..Molecular Constants of Inorganic Compounds, Khimiya, Leningrad (1979).
I. I. Zakharov and G. M. Zhidomirov,Zh. Strukt. Khim.,27, No. 4. 3–10 (1986).
J. Z. Gillies, C. W. Gillies. R. D. Suenram, et al...J. Am. Chem. Soc. 110, 7991–7999 (1988).
J. Z. Gillies. C. W. Gillies, R. D. Suenram. et al.,ibid.,113. 2412–2421 (1991).
S. Roszak. R. J. Buenker, P. C. Hariharan, et al..Chem. Phys.,147. 13–18 (1990).
I. I. Moiseev.Izv. Ross. Akad. Nauk, Sci: Khim. No. 3. 578–588 (1995).
D. L. Bogan. R. S. Sheinson. and F. W. Williams,.J. Am. Chem. Soc,98, 1034–1039 (1976).
N. N. Semenov,On Chemical Kinetics and Reactivity Problems [in Russian], Izd-vo AN SSSR, Moscow (1958).
Author information
Authors and Affiliations
Additional information
Translated fromZhumal Struktumoi Khimii, Vol. 41, No. 2, pp. 240–247,
Rights and permissions
About this article
Cite this article
Tyupalo, N.F., Zakharov, I.I., Kolbasina, O.I. et al. Density functional calculation for ozone-ethylene complexes. J Struct Chem 41, 194–199 (2000). https://doi.org/10.1007/BF02741582
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02741582