Abstract
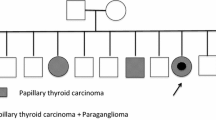
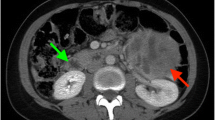
Reports on the association of papillary thyroid carcinoma with paraganglionic or desmoid tumors have appeared infrequently. The former setting usually affects middle-aged females; the latter is typical of familial adenomatous polyposis. We report the case of a 69-yr-old man in whom two abdominal masses had been instrumentally detected following an access of abdominal pain. Save for a moderate hypertension, he was asymptomatic and an impalpable thyroid nodule was detected by ultrasonography. A high urinary noradrenaline output and cytology of the masses raised the suspicion of pheochromocytoma. At laparotomy, an adrenal pheochromocytoma and a paracaval paraganglioma were excised. Subsequently, hemithyroidectomy was performed, and histopathology revealed papillary microcarcinoma. A nodule of desmoid tumor was also removed from the abdominal wall. An analysis ofRET, APC, andTP53 gene mutations, and ofRET andNTRK1 gene rearrangements, yielded negative results. No in vitro transforming activity was detected in the tumor DNA when assayed in transfection experiments. The lack of a consistent family history also made unlikely the possibility of identifying the putative germline defect by linkage analyses. Should this unusual aggregation of tumors represent a new entity, a number of genetic alterations have now been excluded.
Similar content being viewed by others
References
DeLellis RA. Multiple endocrine neoplasia syndromes revisited. Clinical, morphologic, and molecular features. Lab Invest 72:494–505, 1995.
Choyke PL, Glenn GM, Walter MM, Patronas NJ, Linehan WM, Zbar B. Von Hippel-Lindau disease: genetic, clinical, and imaging features. Radiology 194:629–642, 1995.
Carney JA. The triad of gastric epithelioid leiomyosarcoma, pulmonary chondroma, and functioning extra-adrenal paraganglioma: a five-year review. Medicine 62:159–169, 1983.
Carney JA. Carney complex: the complex of myxomas, spotty pigmentation, endocrine overactivity, and schwannomas. Sem Dermatol 14:90–98, 1995.
Mallette LE. Dominant familial syndromes with endocrine hyperfunction: an additional syndrome? Med Hypoth 38:364–367, 1992.
Apel RL, Asa SL, LiVolsi V. Papillary Hurthle cell carcinoma with lymphocytic stroma. “Warthin-like tumor” of the thyroid. Am J Surg Pathol 19:810–814, 1995.
Borst MJ, VanCamp JM, Peacock ML, Decker RA. Mutational analysis of multiple endocrine neoplasia type 2A associated with Hirschsprung's disease. Surgery 117:386–391, 1995.
Ceccherini I, Hofstra RMW, Luo Y, Stulp RP, Barone V, Stelwagen T, Bocciardi R, Nijven H, Bolino A, Seri M, Ronchetto P, Pasini B, Bozzano M, Buys CHCM, Romeo G. DNA polymorphisms and conditions for SSCP analysis of the 20 exons of the ret protooncogene. Oncogene 9:3025–3029, 1994.
Eng C, Smith DP, Mulligan LM, Healey CS, Zvelebil MJ, Stonehouse TJ, Ponder MA, Jackson CE, Waterfield MD, Ponder BAJ. A novel point mutation in the tyrosine kinase domain of theRET proto-oncogene in sporadic medullary thyroid carcinoma and in a family with FMTC. Oncogene 10:509–513, 1995.
Eng C, Smith DP, Mulligan LM, Nagai MA, Healey CS, Ponder MA, Gardner E, Scheumann GFW, Jackson CE, Tunnacliffe A, Ponder BAJ. Point mutation within the thyrosine kinase domain of theRET protooncogene in multiple endocrine neoplasia type 2B and related sporadic tumours. Hum Mol Genet 3:237–241, 1994.
Wells SA Jr, Chi DD, Toshima K, Dehner LP, Coffin CM, Dowton B, Ivanovich JL, DeBenedetti MK, Dilley WG, Moley JF, Norton JA, Donis-Keller H. Predictive DNA testing and prophylactic thyroidectomy in patients at risk for multiple endocrine neoplasia type 2A. Ann Surg 220:237–250, 1994.
Xue F, Yu H, Maurer LH, Memoli VA, Nutile-McMenemy N, Schuster MK, Bowden DW, Mao J-i, Noll WW. GermlineRET mutations in MEN 2A and FMTC and their detection by simple DNA diagnostic tests. Hum Mol Genet 3:635–638, 1994.
Bongarzone I, Pierotti MA, Monzini N, Mondellini P, Manenti G, Donghi R, Pilotti S, Grieco M, Santoro M, Fusco A, Vecchio G, Della Porta G. High frequency of activation of tyrosine kinase oncogenes in human papillary thyroid carcinoma. Oncogene 4:1457–1462, 1989.
Gaidano G, Ballerini P, Gonz JZ, Inghirami G, Neri A, Newcomb EW, Magrath IT, Knowles DM, Dalla-Favera R. P53 mutations in human lymphoid malignancies: Association with Burkitt's lymphoma and chronic lymphocytic leukemia. Proc Natl Acad Sci USA 8817:5413–5417, 1991.
Groden J, Thliveris A, Samowitz WS, Carlson M, Gilbert L, Albertsen H, Joslyn G, Stecens J, Spirio L, Robertson M, Sargeant L, Krapcho K, Wolff E, Burt R, Hughes JP, Warrington J, McPherson J, Wasmuth J, Le Paslier D, Abderrahim H, Cohen D, Leppert M, White R. Identification and characterization of the familial adenomatous polyposis coli gene. Cell 66:589–600, 1991.
Powell SM, Petersen GM, Krush AJ, Booker S, Jen J, Giardiello FM, Hamilton SR, Vogelstein B, Kinzler KW. Molecular diagnosis of familial adenomatous polyposis. N Engl J Med 329:1982–1987, 1993.
Sipple JH. The association of pheochromocytoma with carcinoma of the thyroid gland. Am J Med 31:163–166, 1961.
Williams ED. A review of 17 cases of carcinoma of the thyroid and phaeochromocytoma. J Clin Pathol 18:288–292, 1965.
Steiner AL, Goodman AD, Powers SR. Study of a kindred with pheochromocytoma, medullary thyroid carcinoma, hyperparathyroidism and Cushing's disease: multiple endocrine neoplasia, type 2. Medicine 47:371–409, 1968.
Decker RA. Expression of papillary thyroid carcinoma in multiple endocrine neoplasia type 2A. Surgery 114:1059–1063, 1993.
Eisenberg AA, Wallerstein H: Pheochromocytoma of the suprarenal medulla (paraganglioma). Arch Pathol 14:818, 1932.
Albores-Saavedra J, Espino Durán M. Association of thyroid carcinoma and chemodectoma. Am J Surg 116:887–890, 1968.
Albores-Saavedra J. Personal communication to L. Scopsi, 1996.
Siegelman SS, Zavod R, Hasson J. Hypernephroma, pheochromocytoma and thyroid carcinoma. J Urol 102:402–405, 1969.
Saito Z, Takada T, Miyamoto S, Uchida K, Takeda R, Katsumi T, Lambana F, Matsubara F. Coexistence of papillary thyroid carcinoma and extraadrenal pheochromocytoma causing brain vascular danage: report of an operated case [Japanese]. Shindan To Chiryo—Diagnosis and Treatment 62:2207–2212, 1974.
Oishi S, Sasaki M, Sato T, Isogai M. Coexistence of MEN 2A and papillary thyroid carcinoma and a recurrent pheochromocytoma 23 years after surgery: report of a case and a review of the Japanese literature. Jpn J Clin Oncol 25:153–158, 1995.
Beaugie JM, Belchetz PE, Brown CL, Frankel RJ, Loyd MH. Report of a family with inherited medullary carcinoma of the thyroid and phaeochromocytoma. Br J Surg 62:264–268, 1975.
Melicow MM. One hundred cases of pheochromocytoma (107 tumors) at the Columbia-Presbyterian Medical Center, 1926–1976. Cancer 40:1987–2004, 1977.
Sata K, Matsuzaki M, Nomoto I, Moriyama K. A case of pheochromocytoma developed after resection of adenocarcinoma of thyroid (in Japanese). Nippon Naika Gakkai Zasshi 67:446, 1978.
Kikuchi M, Okuyama M, Takeda N, Miura K, Mishima H. A case of adrenal pheochromocytoma associated with papillary thyroid carcinoma (in Japanese). Shindan To Chiryo—Diagnosis and Treatment 70:2339–2343, 1982.
Kubo S, Imamura T, Kinoshita S, Tagawa H, Fukui J, Koga H, Hara K, Hirayu S, Izumi M, Nagataki S. A case of paraganglioma diagnosed by131I-metaiodobenzyl guanidine (MIBG) scintigraphy (in Japanese). Nippon Naika Gakki Zasshi 73:1201–1208, 1984.
Yasuda G, Shionoiri H, Hamada K, Umemura S, Hiroto S, Takasaki I, Kaneko Y. Bilateral pheochromocytoma associated with papillary adenocarcinoma of thyroid gland. Report of an unusual case. Endocrinol Jpn 32:399–404, 1985.
Yamaguchi T, Nonaka H, Yamaguchi H, Ichikawa M, Kondo T. A case of pheochromocytoma with a papillary adenocarcinoma of the thyroid gland, presenting remarkable hyperreninangiotensinemia and hypercortisolemia (in Japanese). Naika 57:1189–1192, 1986.
Nakamura H, Koga M, Higa S, Sato B, Noma K, Morimoto Y, Kishimoto S, Nakano E, Sonoda T. A case of von Recklinghausen's disease associated with pheochromocytoma and papillary carcinoma of the thyroid gland. Endocrinol Jpn 34:545–551, 1987.
Nakamura S, Ishiyama M, Sugimoto M, Kosaka J, Watanabe H, Horiya Y, Hara S, Shima H. A case of extraadrenal pheochromocytoma with papillary thyroid carcinoma. Endocrinol Jpn 38:351–356, 1991.
Kido Y, Kuwano H, Yano K, Mori M, Kitamura K. Concurrent adrenal pheochromocytoma and papillary adenocarcinoma of the thyroid in a 20-year old man. Tumori 78:59–62, 1992.
Saldana MJ, Salem LE, Travezan R. High altitude hypoxia and chemodectomas. Hum Pathol 4:251–263, 1973.
Parry DM, Li FP, Strong LC, Carney JA, Schottenfeld D, Reimer RR, Grufferman S. Carotid body tumors in humans: Genetics and epidemiology. J Natl Cancer Inst 68: 573–578, 1982.
Larraza-Hernandez O, Albores-Saavedra J, Benavides G, Hrause LG, Perez-Merizaldi JC, Ginzo A. Multiple endocrine neoplasia. Pituitary adenoma, multicentric papillary thyroid carcinoma, bilateral carotid body paraganglioma, parathyroid hyperplasia, gastric leiomyoma, and systemic amyloidosis. Am J Clin Pathol 78:527–532, 1982.
Cayot F, Bastien H, Justrabo E, Mottot C, Cuisenier J, Bruchon Y, Cabanne F. Paragangliomes multiples du cou avec localisation thyroïdienne. Cancer papillaire thyroïdien et adenome parathyroïdien associés. Sem Hôp (Paris) 58:2004–2007, 1982.
Derchi LE, Serafini G, Rabbia C, De Albertis P, Solbiati L, Candiani F, Musante F, Bertoglio C, Rizzatto G. Carotid body tumors: US evaluation. Radiology 182:457–459, 1992.
Barbot N, Calmettes C, Schuffenecker I, Saint-André JP, Franc B, Rohmer V, Jallet P, Bigorgne JC. Pentagastrin stimulation test and early diagnosis of medullary thyroid carcinoma using an immunoradiometric assay of calcitonin: comparison with genetic screening in hereditary medullary thyroid carcinoma. J Clin Endocrinol Metab 78:114–120, 1994.
Lax SF, Beham A, Kronberger-Schönecker D, Langsteger W, Denk H. Coexistence of papillary and medullary carcinoma of the thyroid gland—mixed or collision tumour? Clinicopathologic analysis of three cases. Virchows Arch 242:441–447, 1994.
Scopsi L, Collini P, Sampietro G, Boracchi P, Pilotti S. Prognostic impact of thyroid lymphocytic infiltration in patients with medullary thyroid carcinoma. Thyroid 6:613–617, 1996.
Lamberg B-A, Reissel P, Stenman S, Koivuniemi A, Ekblom M, Mäkinen J, Franssila K. Concurrent medullary and papillary thyroid carcinoma in the same thyroid lobe and in siblings. Acta Med Scand 209:421–424, 1981.
Gagel RF.ret protooncogene mutations and endocrine neoplasia—A story intettwined with neural crest differentiation. Endocrinology 137:1509–1511, 1996.
Eng C, Crossey PA, Mulligan LM, Healey CS, Houghton C, Prowse A, Chew SL, Dahia PLM, O'Riordan LHO, Toledo SPA, Smith DP, Maher ER, Ponder BAJ. Mutations in the RET proto-oncogene and the von Hippel-Lindau disease tumour suppressor gene in sporadic and syndromic phacochromocytomas. J Med Genet 32:934–937, 1995.
Sugg SL, Zheng L, Rosen IB, Freeman JL, Ezzat S, Asa SL.ret/PTC-1,-2, and-3 oncogene rearrangements in human thyroid carcinomas: implications for metastatic potential? J Clin Endocrinol Metab 81:3360–3365, 1996.
Bongarzone I, Fugazzola L, Vigneri P, Mariani L, Mondellini P, Pacini F, Basolo F, Pinchera A, Pilotti S, Pierotti MA. Age-related activation of the tyrosine kinase receptor proto-oncogenes RET and NTRK1 in papillary thyroid carcinoma J Clin Endocrinol Metab81:2006–2007, 1996.
Hayes HM, Fraumeni JF. Chemodectomas in dogs: epidemiologic comparison with man. J Natl Cancer Inst 52:1455–1458, 1974.
Bell B, Mazzaferri EL. Familial adenomatous polyposis (Gardner's syndrome) and thyroid carcinoma. Dig Dis Sci 38:185–190, 1993.
Gurbuz AK, Giardiello FM, Petersen GM, Krush AJ, Offerhaus GJA, Booker SV, Kerr MC, Hamilton SR. Desmoid tumours in familial adenomatous polyposis. Gut 35:377–381, 1994.
Harach HR, Williams GT, Williams ED. Familial adenomatous polyposis associated thyroid carcinoma: a distinct type of follicular cell neoplasms. Histopathology 25:549–561, 1994.
Pilotti S, Collini P, Manzari A, Marubini E, Rilke F. Poorly differentiated forms of papillary thyroid carcinoma: distinctive entities or morphological patterns? Sem Diagn Pathol 12:249–255, 1995.
Miyaki M, Konishi M, Kikuchi-Yanoshita R, Enomoto M, Tanaka K, Takahashi H, Muraoka M, Mori T, Konishi F, Iwama T. Coexistence of somatic and germ-line mutations of APC gene in desmoid tumors from patients with familial adenomatous polyposis. Cancer Res 53:5079–5082, 1993.
Sen-Gupta S, Van der Liujt RB, Bowles LV, Meera Khan P, Delhanty JDA. Somatic mutation of APC gene in desmoid tumour in familial adenomatous polyposis. Lancet 342:552–553, 1993.
Palmirotta R, Curia MC, Esposito DL, Valanzano R, Messerini L, Ficari F, Brandi ML, Tonelli F, Mariani-Costantini R, Battista P, Cama A. Novel mutations and inactivation of both alleles of the APC gene in desmoid tumors. Hum Mol Genet 4:1979–1981, 1995.
Eccles DM, van der Luijt R, Breukel C, Bullman H, Bunyan D, Fisher A, Barber J, du Boulay C, Primrose J, Burn J, Fodde R. Hereditary desmoid disease due to a frameshift muration at codon 1924 of theAPC gene. Am J Hum Genet 59:1193–1201, 1996.
Scott RJ, Froggatt NJ, Trembath RC, Evans DGR, Hodgson SV, Maher ER. Familial infiltrative fibromatosis (desmoid tumours) (MIM 135290) caused by a recurrent 3' APC gene mutation. Hum Mol Genet 5:1921–1925, 1996.
Birch JM. Li-Fraumeni syndrome. Eur J Cancer 30A:1935–1941, 1994.
Greenblatt MS, Bennett WP, Hollstein M, Harris CC. Mutations in the p53 tumor suppressor gene: clues to cancer etiology and molecular pathogenesis. Cancer Res 54:4855–4878, 1994.
Dahia PLM, Aguiar RCT, Tsanaclis AM, Bendit T, Bydlowski SP, Abelin NMA, Toledo SPA. Molecular and immunohistochemical analysis of P53 in phaeochromocytoma. Br J Cancer 72:1211–1213, 1995.
Wang DG, Johnston CF, Anderson N, Sloan JM, Buchanan KD. Over-expression of the tumour suppressor gene p53 is not implicated in neuroendocrine tumour carcinogenesis. J Pathol 175:397–401, 1995.
Yana I, Nakamura T, Shin E, Karakawa K, Kurahashi H, Kurita Y, Kobayashi T, Mori T, Nishisho I, Takai S. Inactivation of the p53 gene is not required for tumorigenesis of medullary thyroid carcinoma or pheochromocytoma. Jpn J Cancer Res 83:1113–1116, 1992.
Sutton MStJ, Sheps SG, Lie JI. Prevalence of clinically unsuspected pheochromocytoma. Review of a 50-year autopsy series. Mayo Clinic Proc 56:354–360, 1981.
Zbaren P, Lehmann W. Carotid body paraganglioma with metastases. Laryngoscope 95:450–454, 1985.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scopsi, L., Cozzaglio, L., Collini, P. et al. Concurrent pheochromocytoma, paraganglioma, papillary thyroid carcinoma, and desmoid tumor: A case report with analyses at the molecular level. Endocr Pathol 9, 79–90 (1998). https://doi.org/10.1007/BF02739955
Issue Date:
DOI: https://doi.org/10.1007/BF02739955