Abstract
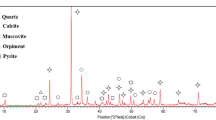
Gold is present in refractory sulfide gold ores mainly in arsenian pyrite and arsenopyrite, where it occurs in both the chemically bonded state and as nano-size grains of metallic gold. During roasting or pressure oxidation, the sulfide matrix is destroyed and essentially all the gold is converted to the metallic form. The liberated gold is readily dissolved in conventional cyanide media, although a few residual gold particles, commonly encapsulated by films of silica-rich gel or by calcium sulfate, are detected. Chloridecontaining gold ores can generate soluble gold chlorocomplexes during hydrometallurgical processing and the dissolved gold can be sequestered on the surfaces of associated carbon particles, resulting in reduced gold recoveries.
Similar content being viewed by others
References
L.J. Cabri et al., “The Nature of ’Invisible’ Gold in Arsenopyrite,”Canadian Mineralogist, 27 (1989), pp. 353–362.
F.E. Wagner, P. Marion, and J.R. Regnard, “Mössbauer Study of the Chemical State of Gold in Gold Ores,”GOLD 100, Proc. Int. Conf. Gold 2—Extractive Metallurgy of Gold (Johannesburg, S.A.: S. Afr. Inst. Min. Metall., 1986), pp. 435–443.
F.J. Arriagada and K. Osseo-Asare, “Gold Extraction from Refractory Ores: Roasting Behavior of Pyrite and Arsenopyrite,”Precious Metals: Mining, Extraction and Processing, ed. V. Kudryk, D.A. Corrigan, and W.W. Liang (Warrendale, PA: The Metallurgical Society of AIME, 1984), pp. 367–385.
H.D. Freeman, J.N. Hartley, and O.J. Wick, “Scanning Electron Microscopic and Microprobe Analysis of Refractory Gold Ores and Process Implications of the Analysis,”Process Mineralogy, ed. D.M. Hausen and W.C. Park (Warrendale, PA: The Metallurgical Society of AIME, 1981), pp. 305–323.
L.J. Cabri et al., “Chemical Speciation of Gold in Arsenopyrite,”Canadian Mineralogist, 38 (2000), pp. 1265–1281.
A.D. Genkin et al., “A Multidisciplinary Study of Gold in Arsenopyrite from Four Mesothermal Gold Deposits in Siberia, Russian Federation,”Economic Geology, 93 (1998), pp. 463–487.
G. Simon et al., “Oxidation State of Gold and Arsenic in Gold-Bearing Arsenian Pyrite,”Am. Mineralogist, 84 (1999), pp. 1071–1079.
G.L. Simmons et al., “Pressure Oxidation Problems and Solutions: Treating Carbonaceous Gold Ores Containing Trace Amounts of Chlorine (Halogens),”Mining Engineering, 50 (1) (1998), pp. 69–73.
P.M. Swash and A.J. Monhemius, “Hydrothermal Precipitation from Aqueous Solution Containing Iron(III), Arsenate and Sulphate,”Hydrometallurgy’94 (New York: Chapman and Hall, 1994), pp. 177–190.
P.A. Riveros, J.E. Dutrizac, and P. Spencer, “Arsenic Disposal Practices in the Metallurgical Industry,”Canadian Metallurgical Quarterly, 40 (2001), pp. 395–420.
T.T. Chen and J.E. Dutrizac, “Characterization of Arsenic-Bearing Wastes Generated in Refractory Gold Processing Operations,”Recycling and Waste Treatment in Mineral and Metal Processing: Technical and Economic Aspects, Vol. 2, ed. B. Bjorkman, C. Samuelsson, and J.-O. Wikstrom (Luleå, Sweden: Luleå University of Technology, 2002), pp. 665–676.
T.T. Chen and J.E. Dutrizac, “A Mineralogical Study of the Cyanide Leach Residues from Pressure-Oxidized Twin Creeks Gold Ore,”EPD Congress 2000, ed. P.R. Taylor (Warrendale, PA: TMS, 2000), pp. 561–577.
J.B. Hiskey, X.H. Jiang, and R. Ramadorai, “Fundamental Studies on the Loading of Gold on Carbon in Chloride Solution,”Gold ’90, ed. D. M. Hausen et al. (Littleton, CO: Society for Mining, Metallurgy and Exploration, 1990), pp. 369–376.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chen, T.T., Cabri, L.J. & Dutrizac, J.E. Characterizing gold in refractory sulfide gold ores and residues. JOM 54, 20–22 (2002). https://doi.org/10.1007/BF02709181
Issue Date:
DOI: https://doi.org/10.1007/BF02709181