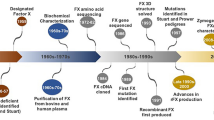
Abstract
Factor VIII (FVIII) functions as a co-factor in the blood coagulation cascade for the proteolytic activation of factor X by factor IXa. Deficiency of FVIII causes hemophilia A, the most commonly inherited bleeding disorder. This review highlights current knowledge on selected aspects of FVIII in which both the scientist and the clinician should be interested.
Similar content being viewed by others
Abbreviations
- FVIII:
-
Factor VIII
- ER:
-
endoplasmic reticulum
- ITI:
-
immune tolerance induction
- vWF:
-
von Willebrant factor
References
Abshire T C, Brackmann H H, Scharrer I, Hoots K, Gazengel C, Powell J S, Goring E, Kellermann E and Voshurgh E 2000 Sucrose formulated recombinant human antihemophilic factor VIII is safe and efficacious for treatment of hemophilia A in home therapy;Thromb. Haemost. 38 811–816
Antonarakis S E, Kazazian H H and Tuddenham E G 1995 Molecular etiology of factor VIII deficiency in hemophilia A;Hum. Mutat. 5 1–22
Antonarakis S E, Rossiter J P, Young M, Horst J, Demoerloose P, Sammer S S, Ketterling R P and Kazazian H H Jr, Negrier C, Vinciguerra C 1995 Factor VIII gene inversions in severe hemophilia A: results of an international consortium study;Blood 86 2206–2212
Azzi A, De Santis R, Morfini M, Zakrzewska K, Musso R, Santagostina E and Castaman G 2001 TT virus contaminates first generation recombinant factor VIII concentrates;Blood 98 2571–2573
Azzi A, Morfini M and Mannucci P M 1999 The transfusion associated transmission of parvovirus B19;Transfus. Med. Rev. 13 194–204
Bowen D J 2002 Haemophilia A and Haemophilia B: molecular insights;Mol. Pathal. 55 127–144
Dimichele D M 1998 Immune tolerance: a synopsis of the international experience;Haemophilia 4 568–573
Eaton D, Rodriguez H and Vehar G A 1986 Proteolytic processing of human factor VIII. Correlation of specific cleavages by thrombin, factor Xa, and activated protein C with activation and inactivation of factor VIII coagulant activity;Biochemistry 25 505–512
Foster P A and Zimmerman T S 1989 Factor VIII structure and function;Blood Rev. 3 180–191
Fricke W A and Lamb M A 1993 Viral safety of clotting factors;Semin. Thromb. Hemost. 19 54–61
Fulcher C A, Roberts J R and Zimmerman T S 1983 Thrombin proteolysis of purified factor VIII. Corelation of activation with generation of a specific polypeptide;Blood 61 807–811
Ghirardini A, Puopolo M, Chiarotti F and Mariani G 1996 The international registry of immune tolerance: 1994; Update;Vox Sang. 70 42–46
Gilles J G, Lavend’homme R, Peerlinck K, Jacquemin M G, Hoxlaerts M, Joorieux S, Mazurier C, Vermylen J and Saint-Remy J M 1999 Some factor VIII (FVIII) inhibitors recognize a FVIII epitope(s) that is present only on FVIII - vWF complexes;Thromb. Haemost. 82 40–45
Gitschier J, Wood W I, Goralka T M, Wion K L, Chen E Y, Eaton D H, Vehar G A, Capon D J and Lawn RM 1984 Characterization of the human factor VIII gene;Nature (London) 312 326–330
Hoeben R C, Einerhand M P W, Briet E, Van Ormondt H, Valerio D and Van der Eb A J 1992 Towards gene therapy in hemophilia A: retrovirus-mediated transfer of a factor VIII gene into murine haemotopoietic progenitor cells;Thromb. Haemost. 67 341–345
Hoeben R C, Fallaux F J, Van Tilburg N H, Cramer S J, Van Briet E and Van Der Eb A J 1993 Towards gene therapy of hemophilia A: long term persistence of Factor VIII secreting fibroblasts after transplantation into immunodeficient mice;Hum. Gene Ther. 4 179–186
Jacquemin M, Lavend’homme R, Benhida A, Vanzieleghem B, d’Oiron R, Lavergne J M, Brackmann H H, Schwaab R, Vanden Driessche T, Chuah M K L, Hoylaert S M, Gilles J G G, Peerlink K, Vermylen J and Saaint-Remy J M R 2000 A novel cause of mild/moderate hemophilia A: Mutations scattered in the factor VIII C1 domain reduce factor VIII binding to von Willebrand factor;Blood 96 958–965
Kaufman R J 1992 Biological regulation of factor VIII activity;Annu. Rev. Med. 43 325–339
Kaufman R J 1999 Advances toward gene therapy for hemophilia at the milennium;Hum. Gene Ther. 10 2091–2107
Kreil T R, Zimmermann K, Pable S, Schwarz H P, Dorner F, Azzi A and Morfini 2002 TT virus does not contaminate first generation recombinant factor VIII concentrate;Blood 100 2271–2272
Lakich D, Kazazian H H Jr, Antonarakis S E and Gitschier J 1993 Inversions disrupting the factor VIII gene are a common cause of severe haemophilia A;Nat. Genet. 5 236–241
Lenting P J, van Mourik J A and Mertens K 1998 The life cycle of coagulation factor VIII in view of its structure and function;Blood 92 3983–3996
Leyte A, van Schijndel H B, Niehrs C, Hunter W B, Verbeet M P, Mertens K and van Mourik J A 1991 Sulphation of Tyr1680 of human blood coagulation factor VIII is essential for the interaction of factor VIII with von Willebrand factor;J. Biol. Chem. 266 740–746
Lozier J N and Kessler C N 2000 Clinical aspects and therapy of hemophilia; inHematology: basic principles and practice (eds) R Hoffman, E Benz Jr and S Shattil (New York: Churchill Livingstone) pp 1883–1904
Lusher J M, Arkin S, Abildgaard C F and Schwartz R S 1993 Recombinant factor VIII for the treatment of previously untreated patients with haemophilia A - Safety, efficacy, and development of inhibitors;N. Engl. J. Med. 328 453–459
Lusher J M, Petrini P and Angiolillo A 2000 Antibody and inhabitor patterns in previously untreated patients (PUPs) treated exclusively with B-domain deleted factor VIII (BDDrFVIII);Blood 96 266
Lynch C M, Israel D I, Kaufman R J and Miller D 1993 Sequences in the coding region of clotting factor VIII act as dominant inhibitors of RNA accumulation and protein production;Hum. Gene Ther. 4 259–273
Mannucci P M and Tuddenham E G D 2001 Medical Progress: The haemophilias - from Royal genes to gene therapy;N. Engl. J. Med. 344 1773–1779
Mariani G, Ghirardini A and Bellocco R 1994 Immune tolerance in hemophilia - Principal results from the international registry. Report of the factor VIII and IX subcommittee;Thromb. Haemost. 72 155–158
Marquette K A, Pittman D D and Kaufman R J 1995 A 110 amino acid region within the A1-domain of coagulation factor VIII inhibits secretion from mammalian cells;J. Biol. Chem. 270 10297–10303
Martin R B 1986Metal ions in biological system (ed.) H Segel (New York: Marcel Dekker) pp 20–21
Matsui H, Sugimoto M, Tsuji S, Shima M, Giddings J and Yoshioka A 1999 Transient hypoplastic anemia caused by primary human parvovirus B19 infection in previously untreated patient with hemophilla transfused with a plasma derived monoclonal antibody purified factor VIII concentrate;J. Pediatr. Hematol. Oncol. 21 74–76
Mauser-Bunschoten E P, Roosendaal F R, Nieuwenhuis H P, Rosendaal G, Briet E and van den Berg H M 1994 Clinical course of factor VIII inhibitors developed after exposure to a pasteurised Dutch concentrate compared to classic inhibitors in haemophilia A;Thromb. Haemost. 71 703–706
McEvoy G K, 2001AHFS Drug Information (Bethesda: American Society of Health System Pharmacists) pp 1412–1428
Naylor J, Brinke A, Hassock S, Green P M and Giannelli F 1993 Characteristic mRNA abnormality found in half the patients with severe haemophilia A is due to large DNA inversions;Hum. Mol. Genet. 2 1973–1978
Nishizawa T, Okamoto N, Konishi K, Yoshizawa H, Miyakawa Y and Mayumi M 1997 A novel DNA virus (TTV) associated with elevated transaminase levels in posttransfusion hepatitis of unknown etiology;Biochem. Biophys. Res. Commun. 241 92–97
Osterberg T, Fatouros A and Mikaelsson M 1997 Development of a freeze dried albumin free formulation of recombinant factor VIII SQ;Pharm. Res. 14 892–898
Peerlinck K, Arnout J, di Giampattista M, Gillies J G, Laub R, Jacquemin M, Saint J M R and Vermylen J 1997 Factor VIII inhibitors in previously treated haemophilia A patients with a double virus inactivated plasma derived concentrate;Thromb. Haemost. 77 80–86
Peerlinck K, Arnout J, Gilles J G, Saint-Remy J M and Vermylen J 1993 A higher than expected incidence of factor VIII inhibitor in multitransfused haemophilia A patients treated with a intermediate purity pasteurized factor VIII concentrate;Thromb. Haemost. 69 115–118
Pipe S W, Morris J A, Shah J and Kaufman R J 1998 Differential interaction of coagulation factor VIII and factor V with protein chaperones calnexin and calreticulin;J. Biol. Chem. 273 8537–8544
Pitmann D D and Kaufman R J 1988 Proteolytic requirements for thrombin activation of antihemophilic factor VIII;Proc. Natl. Acad. Sci. USA 85 2429–2433
Pittman D D, Wang J H and Kaufman R J 1992 Identification and functional importance of tyrosine sulfate residues within recombinant factor VIII;Biochemistry 31 3315–3325
Richard K A 1997 The diagnosis of hemophilia A and B and von Willebrand’s disease; inHemophilia (eds) C D Forbes, L M Aledort and R Madhok (London: Chapman and Hall Medical) pp 53–62
Roelse J C, De Laaf R T, Timmermans S M, Peters M, Van Mourik J A and Voorberg J 2000 Intracellular accumulation of factor VIII induced by missense mutations Arg 593 - Cys and Asn 618 - Ser explains cross-reacting material reduced haemophilia A;Br. J. Haematol. 108 241–246
Rosendaal F R 1997 Factor VIII inhibitors on a SD treated and pasteurised concentrate associated with specific batches and batch characteristics;Thromb. Haemost. 78 590–594
Rosendaial F R, Nieuwenhuis H K, van den Berg H M, Heijboer H, Mauser-Bunschoten E P, van der Meer J, Smit C, Strengers P F and Briet E 1993 A sudden increase in factor VIII inhibiter development in multitransfused hemophilia A patients in the Netherlands. Dutch Hemophilia study Group;Blood 81 2180–2186
Roth D A, Tawa N E Jr, O’Brien J M, Treco D A and Selden R F 2001 Nonviral transfer of the gene encoding coagulation factor VIII in patients with severe Hemophilia A;N. Engl. J. Med. 344 1735–1742
Saenko E L and Scandella D 1997 The acidic region of the factor VIII light chain and the C2 domain together from the high affinity binding site for von Willebrand factor;J. Biol. Chem. 272 18007–18014
Saenko E L, Shima M, Rejalakshmi K J and Scandella D 1994 A role for the C2 domain of factor VIII in binding to von Willebrand factor;J. Biol. Chem. 269 11601–11605
Scandella D 2000 Properties of anti factor VIII inhibitor antibodies in hemophilia A patients;Semin. Thromb. Hemost. 26 137–142
Schwaab R, Brackmann H H, Meyer C, Seehafer J, Kirchgesser M, Hacck A, Olek K, Tuddenham E G D and Oldenburg J 1995 Haemophilia A: Mutation type determines risk of inhibitor formation;J. Thromb. Haemost. 74 1402–1406
Srivastava A, Chuansumrit A, Chandy M, Duraiswamy G and Karagus C 1998 Management of hemophilia in the developing countries;Hemophilia 4 474–480
Swaroop M, Moussalli M, Pipe S W and Kaufman R J 1997 Mutagenesis of a potential immunoglobulin-binding-protein-binding site enhances secretion of coagulation factor VIII;J. Biol. Chem. 272 24121–24124
Tuddenham E G D, Cooper D N, Gritschier J, Higuchim, Hoyer L W, Yoshioka A, Peake I R, Schwaab R, Olekk, Kazazian H H 1991 Haemophilia A: database of nucleotide substitutions, deletions, insertions and rearrangements of the factor VIII gene;Nucleic Acids Res. 19 4821–4833
Vehar G A, Keyt B, Eaton D, Rodriguez H, O’Brien D P, Rotblat F, Oppermann H, Keck R, Wood W I, Harkins R N, Tuddenham E G D, Lawn R M and Capon D J 1984 Structure of human Factor VIII;Nature (London) 312 337–342
Viot A J, Koppelman S J, van den Berg M H, Bouma B N and Sixma J J 1995 The affinity and stoichiometry of binding of human factor VIII to von Willebrand factor;Blood 85 3150–3157
Wacey A I, Kemball-Cook G, Kazazian H H, Antonarakis S E, Schivaab R, Lindey P and Tuddenhum E G 1996 The haemophilia A mutation search test and resource site, home page of the factor VIII mutation database: HAMSTeRS;Nucleic Acids Res. 24 100–102
Wion K L, Kelly D, Summerfield J A, Tuddenham E G D and Lawn R M 1985 Distribution of factor VIII mRNA and antigen in human liver and other tissues;Nature (London) 317 726–729
Yee T T, Lee C A and Pasi K J 1995 Life threatening human parvovirus B19 infection in immunocompetent haemophilia;Lancet 345 794–795
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhopale, G.M., Nanda, R.K. Blood coagulation factor VIII: An overview. J Biosci 28, 783–789 (2003). https://doi.org/10.1007/BF02708439
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02708439