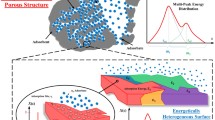
Abstract
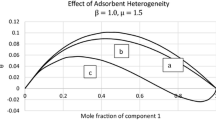
The adsorption isotherms with each saturation vapor pressure factor (cs1, cs2 or cs3) for two groups of sites in two cases of the multilayer and for three groups of sites in one case of the multilayer are derived statistically in heterogeneous non-porous solid adsorbents without interactions among the adsorbed molecules. When some sites of BET isotherm are substituted by less energetic sites, the two-group isotherm obtained by the substitution shows less adsorption over the whole range of relative pressure than the BET isotherm prior to the substitution, at any combined values of f1 with M1 of the two-group isotherm with the same saturation vapor pressure factor. A method to get the monolayer sites (vm) from the ratios of the experimental isotherm to the theoretical isotherm at the whole relative vapor pressure minimizing the standard error is suggested. Our two- or three-group isotherms calculated through many experimental adsorption isotherm data selected appropriately provide larger values of vm than those obtained from BET isotherms. Differential heat vs. v/vm and Bose-Condensation heat are mentioned.
Similar content being viewed by others
References
Adamson, A. W., “Adsorption of Gases and Vapors on Solids,” Physical Chemistry of Surfaces, Chap. 16, John Wiley & Sons. Inc 5th ed., 591 (1990).
Anderson, R. B., “Modification of the Brunauer, Emmett and Teller Equation,”J. Am. Chem. Soc,68, 686 (1946).
Brunauer, S., Deming, L. S., Deming, W. E. and Teller, E., “On Theory ofthe van der Waals Adsorption of Gases,”J. Am. Chem. Soc,62, 1723 (1940).
Brunauer, S., Emmett, P. H. and Teller, E., “Adsorption of Gases in Multimolecular Layers,”J. Am. Chem Soc,60, 309 (1938).
Drain, L. E. and Morrison, J. A., “Thermodynamic Properties of Argon Adsorbed on Rutile,”Trans. Faraday Soc,48, 840 (1952).
Drain, L. E. and Morrison, J. A., “Thermodynamic Properties of Nitrogen and Oxygen Adsorbed on Rutile,”Trans. Faraday Soc,49, 654 (1953).
Gerald, P.,“Modification of the Brunauer-Emmett-Teller Theory of Multimolecular Adsorption,”J. Am. Chem. Soc,67, 1958 (1945).
Gregg, S. J. and Sing, K. S. W., “Physical Adsorption of Gases by Non-porous Solids,” Chapter 2, Adsorption, Surface Area and Porosity, Academic Press Inc.(Ltd.) (1969).
Hackerman, N. and Hall, A. C., “The Adsorption of Water Vapor on Quartz and Calcite,”J. Phys. Chem.,62, 1212 (1958).
Harkins, W D. and Jura, G., “Surfaces of Solids. XII. An Absolute Method for the Determination of the Area of a Finely Divided Crystalline Solid,”J. Am. Chem. Soc,66, 1362 (1944).
Hill, T. L., “Statistical Mechanics of Mutimolecular Adsorption. I,”J. Chem. Phys.,67, 1958 (1946).
Hill, T. L., “Statistical Mechanics of Adsorption. VI, Localized Unimolecular Adsorption on a Heterogeneous Surface,”J. Chem. Phys.,17, 762 (1949).
Hollaraugh, C. M. and Chessick, J. J., “Adsorption of Water and Polar Paraffinic Compounds onto Rutile,”J. Phys. Chem,65, 109 (1961).
Isirikyan, A. A. and Kiselev, A. V., “The Absolute Adsorption Isotherms of Vapors of Nitrogen, Benzene and n-Hexane, and the Heats of Adsorption of Benzene and n-Hexane on Graphitized Carbon Blacks. I. Graphitized Thermal Blacks,”J. Phys. Chem.,65, 601 (1961).
Jaroniec, M. and Madey, R., “Physical Adsorption on Heterogeneous Solids,” Elsevier, Amsterdam etc. (1988).
Joyner, L. G. and Emmett, P. H., “Differential Heats of Adsorption of Nitrogen on Carbon Blacks,”J. Am. Chem. Soc,70, 2353 (1948).
Kim, D., “Statistical Adsorption Isotherms on Heterogeneous Solid Adsorbent and Statistical Cation Distributions on Y Zeolites,” Ph. D. Thesis, Seoul, Hanyang University (2000), expected.
Knuth, E. L., “Perfect Diatomic Gas,” introduction to STATISTICAL THERMODYNAMICS, McGraw Hill, Chapter 13 (1966).
Langmuir, “The Adsorption of Gases on Plane Surfaces of Glass, Mica and Platinum,”J. Am. Chem. Soc,40, 1361 (1918).
Loeser, E. H., Harkins, W. D. and Twiss, S. B., “Molecular Interaction between n-Propyl Alcohol and Iron or Iron Oxides,”J. Phys. Chem.,57, 591 (1953).
McQuarrie, D. A., “Statistical Thermodynamics,” Chap. 4, Harper & Row Publishers, 79 (1973).
Rhodin, T. N. Jr., “Studies of Copper Surfaces by Low Temperature Adsorption Isotherms,”J. Amer. Chem. Soc,72, 4343 (1950).
Rhodin, T. N., “Physical Adsorption on Single Crystal Zinc Surfaces,”J. Phys. Chem.,57, 143 (1953).
Rudizinski, W. and Evertt, D. H., “Adsorption of Gases on Heterogeneous Sufaces,” Academic Press, New York etc. (1992).
Sears, F. W. and Salinger, G. L., “Statistical Thermodynamics,” Thermodynamics, Kinetic Theory, and Statistical Thermodynamics, Chapter 11, Addison-Wesdey Publishing Company, 3rd ed., 302 (1975).
Timmermann, E., “A B.E.T-like Three Sorption Stage Isotherm,“J. Chem. Soc. Faraday Trans. 1,85 (7), 1631 (1989).
Tompkins, F. C., “Physical Adsorption on Non-uniform Surfaces,“Tran. Faraday Soc,46, 569 (1950).
Van Dun, J. J. and Mortier, W. J., “Temperature-Dependent Cation Distribution in Zeolites. 1. Statistical Thermodynamical Model,“J. Phys. Chem.,92, 6740 (1988).
Van Dun, J. J., Dhaeze, K. and Mortier, W. J., “Temperature-Dependent Cation Distribution in Zeolites. 2,”J. Phys. Chem.,92, 6747 (1988).
Zettemoyer, A. C., Chand, A. and Gamble, E., “Sorption by Organic Substances. I. Krypton and Nitrogen on Polyethylene, Nylon and Collagen,”J. Amer. Chem. Soc,72, 2752 (1950).
Author information
Authors and Affiliations
Additional information
An erratum to this article can be found online at http://dx.doi.org/10.1007/s11814-010-0159-x.
Rights and permissions
About this article
Cite this article
Kim, D. Statistically modeled adsorption isotherms for the multilayer gas molecules adsorbed on non-porous solid adsorbents of two and three groups sites. Korean J. Chem. Eng. 17, 156–168 (2000). https://doi.org/10.1007/BF02707137
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707137