Abstract
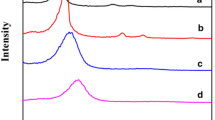
Both powder-type and film-type mesoporous silica materials have been prepared by using a sol-gel process in highly acidic, solvent-rich and mild conditions by solvent evaporation method. Powder mesoporous silica which has hexagonal arrays was well synthesized within only 10 minutes, and mesoporous film was formed on the glass plate within only several seconds by supramolecular interactions between the silicate species and surfactant-templates. In particular, the synthesis time was very short compared with that of hydrothermal synthesis owing to the solvent evaporation. This method has some characteristics different from the hydrothermal method such as the following. There is no precipitation because particles do not form in the solution before solvent evaporation, bulk powder is rigid, whereas that of hydrothermal synthesis is very soft, and mesoporous materials are synthesized in acidic condition. Solvent evaporation method for the synthesis of mesoporous materials is promising because the method is very simple and time-saving.
Similar content being viewed by others
References
Beck, J. S., Vartuli, J. C., Roth, W. J., Leonowicz, M. E., Kresge, C. T., Schmitt, K. T., Chu, C. T.-W., Olson, D. H., Sheppard, E. W., McCullen, S. B., Higgins, J. B. and Schlenker, J. L., “A New Family of Mesoporous Molecular Sieves Prepared with Liquid Crystal Templates,”J. Am. Chem. Soc.,114, 10834 (1992).
Brinker, C. J., Hurd, A. J., Schunk, P. R., Frye, G. C. and Ashley, C. S., “Review of Sol-Gel Thin Film Formation,”J. Non-Cryst. Solids,147,148, 424 (1992).
Bruinsma, P. J., Kim, A. Y., Liu, J. and Baskaran, S., “Mesoporous Silica Synthesized by Solvent Evaporation: Spun Fibers and Spray-Dried Hollow Spheres,”Chem. Mat.,9, 2507 (1997).
Calabro, D. C., Valyocsik, E. W. and Ryan, F. X.,“ In situ ATR/ FTIR Study of Mesoporous Silicate Syntheses,”Microporous Mater.,7, 243 (1996).
Estermann, M., McCusker, L. B., Baeriocher, C., Merroche, A. and Kessler, H., “A Synthetic Gallophosphate Molecular-Sieve with a 20-Tetrahedral-Atom Pore Opening,”Nature,352, 320 (1991).
Huo, Q., Margolese, D. I., Ciesla, U., Demuth, D. G., Feng, P., Gier, T. E., Sieger, P., Firouzi, A., Chmelka, B. F., Schuth, F. and Stucky, G. D., “Organization of Organic Molecules with Inorganic Molecular Species into Nanocomposite Biphase Arrays,”Chem. Mat.,6, 1176 (1994a).
Huo, Q., Margolese, D. I., Ciesla, U., Feng, P., Gier, T. E., Sieger, P., Leon, R., Petroff, P. M., Schuth, F. and Stucky, G. D., “Generalized Synthesis of Periodic Surfactant/Inorganic Composite Materials,”Nature,368, 317 (1994b).
Kim, C.-H. and Woo, S. I., “A Study on the Optimum Synthesis of Mesoporous Molecular Sieve, MCM-41,”HWAHAK KONGHAK,35, 135 (1997).
Kresge, C. T., Leonowicz, M. E., Roth, W. J., Vartuli, J. C. and Beck, J. S., “Ordered Mesoporous Molecular Sieves Synthesized by a Liquid-Crystal Template Mechanism,”Nature,359, 710 (1992).
Lu, Y., Ganguli, R., Drewien, C. A., Anderson, M. T., Brinker, C. J., Gong, W., Guo, Y., Soyez, H., Dunn, B., Huang, M. H. and Zink, J. I., “Continuous Formation of Supported Cubic and Hexagonal Mesoporous Films by Sol-Gel Dip-Coating,”Nature,389, 364 (1997).
Nishida, F., McKiernan, J. M., Cunn, B., Zink, J. I., Brinker, C. J. and Hurd, A. J., “In Situ Fluorescence Probing of the Chemical Changes during Sol-Gel Thin Film Formation,”J. Am. Ceram. Soc.,78, 1640 (1995).
Ogawa, M. and Kikuchi, T., “Preparation of Self-Standing Transparent Films of Silica-Surfactant Mesostructured Materials and the Conversion to Porous Silica Films,”Adv. Mat.,10, 1077 (1998). Park, S.-E., Kim, D. S., Chang, J.-S. and Kim, W. Y., “Photoluminescence Spectroscopic Monitoring in the Synthesis of Mesoporous Materials by Microwave-Induced Heating,”Stud. Surf. Sci. Catal.,117, 265 (1998a).
Park, S.-E., Kim, D. S., Chang, J.-S. and Kim, W. Y., “Synthesis of MCM-41 Using Microwave Heating with Ethylene Glycol,”Catal. Today,44, 301 (1998b).
Ryoo, R., Ko, C. H. and Howe, R. F., “Imaging the Distribution of Framework Aluminum in Mesoporous Molecular Sieve MCM41,”Chem. Mater.,9, 1607 (1997a).
Ryoo, R., Ko, C. H., Cho, S. J. and Kim, J. M., “Optically Transparent, Single-Crystal-Like Oriented Mesoporous Silica Films and Plates,”J. Phys. Chem. B,101, 10610 (1997b).
Sayari, A., “Catalysis by Crystalline Mesoporous Molecular-Sieves,”Chem. Mater.,8, 1840 (1996).
Stucky, G. D., Huo, Q., Firouzi, A., Chmelka, B. F., Schacht, S., Voigt-Martin, I. G. and Schuth, F., “Directed Synthesis of Organic/Inorganic Composite Structures,”Stud. Surf. Sci. Catal.,105, 3 (1997).
Wu, C.-G. and Bein, T., “Microwave Synthesis of Molecular Sieve MCM-41,”Chem. Commun., 925 (1996).
Yang, H., Coombs, N., Sokolov, I. and Ozin, G. A., “Free-Standing and Oriented Mesoporous Silica Films Grown at the Air-Water Interface,”Nature,381, 589 (1996a).
Yang, H., Kuperman, A., Coombs, N., Mamiche-Afara, S. and Ozin, G. A., “Synthesis of Oriented Films of Mesoporous Silica on Mica,”Nature,379, 703 (1996b).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roh, HS., Chang, JS. & Park, SE. Synthesis of mesoporous silica in acidic condition by solvent evaporation method. Korean J. Chem. Eng. 16, 331–337 (1999). https://doi.org/10.1007/BF02707121
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707121