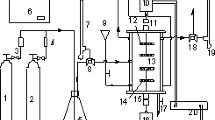
Abstract
In our recent studies, absorption of H2S into the aqueous Fe2(SO4)3 and FeCl3 solutions with various values of ionic strength and pH was investigated in an agitated vessel with a flat interface. The experimental results for both systems revealed that the species which reacts with H2S is FeOH2+. The absorption rates were explained by the theory of gas absorption with an irreversible (l,l)-th order reaction between H2S and FeOH2+. The reaction rate constants were independent of the ionic strength of the solution and correlated as a function of temperature. In this paper, kinetics and mechanism of these absorption reactions are reviewed and the identity for both systems is emphasized.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Asai, S., Konishi, Y. and Yabu, T., “Kinetics of Absorption of Hydrogen Sulfide into Aqueous Ferric Sulfate Solutions”,AIChE J.,36, 1331 (1990).
Asai, S., Nakamura, H. and Aikawa, H., “Absorption of Hydrogen Sulfide into Aqueous Ferric Chloride Solutions”,J. Chem. Eng. Japan,30, 500 (1997).
Dean, J. A., “Lange’s Handbook of Chemistry”, McGraw-Hill, New York (1985).
Hikita, H. and Asai, S., “Gas Absorption with (m,n)-th Order Irreversible Chemical Reaction”,Kagaku Kogaku,27, 823 (1963).
Hikita, H., Asai, S. and Ishikawa, H., “LévÊque Model for Mass Transfer with an Irreversible Second-Order Chemical Reaction”,Bull. Univ. Osaka Pref.,All, 57 (1973).
Hikita, H., Asai, S., Ishikawa, H. and Saito, Y., “Kinetics of Absorption of Chlorine in Aqueous Acidic Solutions of Ferrous Chloride”,Chem. Eng. Sci.,30, 607 (1975).
Imaizumi, T., “Some Industrial Applications of Inorganic Microbial Oxidation in Japan”,Biotech. Bioeng. Symp.,16, 363 (1986).
Kikuchi, T, Komori, N., Noguchi, H., Honna, K. and Iida, H., “A New Process for Hydrogen and Sulfur Recovery from Hydrogen Sulfide”, Preprints I.: Sessions 1.1-5.2, 3.7-25, 4th World Congress of Chemical Engineering, Karlsruhe, Germany (1991).
Smith, R. M. and Martell, A. E., “Critical Stability Constants”, Inorganic Complexes, Plenum Press, New York, 4 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Asai, S., Nakamura, H. & Konishi, Y. Kinetics of absorption of hydrogen sulfide into aqueous Fe(III) solutions. Korean J. Chem. Eng. 14, 307–311 (1997). https://doi.org/10.1007/BF02707044
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02707044