Abstract
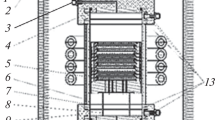
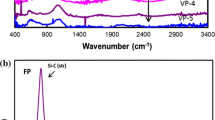
We have used rapid thermal chemical vapor deposition (RTCVD) technique to grow epitaxial SiC thin films on Si wafers without carbonization process by pyrolyzing tetramethylsilane (TMS). The growth rate of SiC films increases with TMS flow rate and temperature, but it decreases with temperature at higher TMS flow rates. The XRD spectra of the films indicate that the growth direction is along the (111) direction of β-SiC. IR and RBS measurements have been employed to analyze the chemical composition of the films. At 1100°C TMS molecules dissociate almost completely into Si atoms, CH4 and C2H2 gases. The growth mechanism of SiC films on Si substrates without carbonization process has been proposed based on the analyses by TEM and QMS.
Similar content being viewed by others
References
Avigal, Y., Schieber, M. and Levin, R.,“Growth of Heteroepitaxial SiC Films by Pyrolysis of Various Alkyl-Silicon Compounds”,J. Cry st. Growth,24/25, 188 (1974).
Chiu, C. C., Desu, S. B. and Tai, C. Y.,“Low Pressure Chemical Vapor Deposition (LPCVD) of Β-SiC on Si(100) Using MTS in a Hot Wall Reactor”,J. Mater. Res.,8, 2617 (1993).
Davis, R. F., Keiner, G., Shur, M., Palmour, J. W. and Edmond, J. A.,“Thin Film Deposition and Microelectronic and Optoelectronic Device Fabrication and Characterization in Monocrystalline Alpa and Beta Silicon Carbide ”,Proceedings of the IEEE,79, 677 (1991).
Figueras, A., Garelik, S., Rodriguez-Clemente, R., Armas, B., Combescure, C. and Dupuy, C.,“A Morphological and Structural Study of SiC Layers Obtained by LPCVD Using Tetramethylsilane”,J. Cryst. Growth,110, 528 (1991).
Golecki, I., Reidinger, F. and Marti, J.,“Single-crystalline, Epitaxial Cubic SiC Films Grown on Si (100)Si at 750‡C by Chemical Vapor Deposition”,Appl Phys. Leu.,60, 1703 (1992).
Herlin, N., Lefebvre, M., Pealat, M. and Perrin, J., Investigation of the Chemical Vapor Deposition of Silicon Carbide from Tetramethylsilane by In-situ Temperature and Gas Composition Measurements”,J Phys. Chem.,96, 7063 (1992).
Ho, P. and Breiland, W. G.,“Observation of Gas-Phase Si Atoms in the Chemical Vapor Deposition of Silicon from Dichlorosilane”,J Appl. Phys.,63, 5184 (1988).
Jacobson, K. A.,“Growth, Texture, and Surface Morphology of SiC Layers”,J. Electrochem. Soc. 118, 1001 (1971).
Maya, L.,“Deposition of Amorphous Hydrogenated Silicon Carbide Films Using Organosilanes in an Argon/Hydrogen Plasma”,J. Vac. Sci. Technol.,A12, 754 (1994).
Mogab, C. J. and Leamy, H. J.,“Conversion of Si to Epitaxial SiC by Reaction with C2H2”,J. Appl. Phys.,45, 1075 (1974).
Motoyama, S.-I., Morikawa, N. and Kaneda, S.,“Low-Temperature Growth and Its Growth Mechanisms of 3C-SiC Crystal by Gas Source Molecular Beam Epitaxial Method”,J. Cryst. Growth,100, 615 (1990).
Nishino, S., Suhara, H., Ono, H. and Matsunami, H.,“Epitaxial Growth and Electric Characteristics of Cubic SiC on Silicon”,J. Appl. Phys.,61, 4889 (1987).
Powell, J. A. and Will, H. A., Recent Developments in SiC, in Springer Proceedings in Physics, vol. 34, Amorphous and Crystalline Silicon Carbide, Springer-Verlag Berlin, Heidelberg, 1989.
Schlichting, J.,“Chemical Vapor Deposition of Silicon Carbide”,Powder Matall. Int.,12, 141 (1980).
Steckl, A. J. and Li, J. P.,“Epitaxial Growth of Β-SiC on Si by RTCVD with C3H8 and SiH4”,IEEE Trans. Electron Device,39, 64 (1992).
Stenhagen, E., Abrahamsson, S. and McLafferty, F., Atlas of Mass Spectral Data”, John Wiley & Sons, New York, 1969.
Takahashi, K., Nishino, S. and Saraie, J.,“Low Temperature Growth of 3C-SiC on Si Substrate by Chemical Vapor Deposition Using Hexamethyl-disilane as a Source Material”,J. Electrochem. Soc,139, 3565 (1992).
Veintemillas, S., Madigou, V., Rodriguez-Clemente, R. and Figueras, A.,“Thermodynamic Analysis of Metalorganic Chemical Vapor Deposition of SiC Using Tetramethylsilane as Precursor”,J. Cryst. Growth,148, 383 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Seo, Y.H., Nahm, K.S., Suh, E.K. et al. Growth mechanism of 3C-SiC(lll) on Si without carbonization process. Korean J. Chem. Eng. 13, 522–529 (1996). https://doi.org/10.1007/BF02706004
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02706004