Abstract
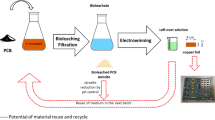
The possibility of using electrolytic processing to regenerate etchants while simultaneously recovering copper used in the printed-wiring-board industry is considered for such systems as ferric chloride, acid, and ammoniacal-cupric chloride. This article discusses the cell design and anodic and cathodic processes, and concludes that the electrolytic process is ecofriendly with a minimal discharge of effluents.
Similar content being viewed by others
References
G.N. Srinivasan et al., “Electrolytic Recovery of Copper with Simultaneous Regeneration of Ferric Chloride Etchants” (Paper presented at “Advances in Electrochemical Science and Technology” held at IISC, Bangalore, July 1999).
P. Eisler,The Technology of Printed Circuit (London: Heywood, 1962), pp. 223–231.
MR. Hills, “The Electrolytic Regeneration of Spent Ferric Chloride Etchant,”Trans. IMF, 57 (1979), p. 73.
Technical Report of Tristar, Methuen, Massachusetts.
Technical Report of Tristar, Methuen, Massachusetts.
Technical Report of FSL, U.K., 1989.
Technical Report of Elochem, Germany.
G.N. Srinivasan et al., “Electrolytic Recovery of Copper and Regeneration of Cupric Chloride Etchant,”Trans. IIM, 51 (1998), pp. 379–382.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Adaikkalam, P., Srinivasan, G.N. & Venkateswaran, K.V. The electrochemical recycling of printed-wiring-board etchants. JOM 54, 48–50 (2002). https://doi.org/10.1007/BF02701851
Issue Date:
DOI: https://doi.org/10.1007/BF02701851