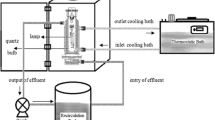
Abstract
Experiments were performed to investigate the effect of operating parameters on the photodegradation efficiency of paraquat in a TiO2-suspended photoreactor with immersed UV lamps. TiO2 particles were prepared by a hydrothermal method. The removal rate of paraquat in the reactor was 0.54 mg/l/h with only air-sparging. The removal rate in 24 h with both the UV radiation and air-sparging was 50% higher than that with only the UV radiation. Variations of the paraquat concentration at the UV intensities of 4 and 8 W/m2 decreased slowly with time, but that at 12 W/m2 decreased more rapidly. The removal efficiency at the air-sparging flow rate of 1 //min increased as a UV light intensity increased. pH value in the reactor at the UV intensity of 12 W/m2 decreased with time until 12 h and then increased with time over 12 h.
Similar content being viewed by others
References
Boukas, T. and Mantzouridou, F., “Effect of the Aeration Rate on Pullulan Production and Fermentation Broth Rheological Properties in an Airlift Reactor”,J. Chem. Technol. Biotechnol.,76, 371 (2001).
Braun, S. S., Maurette, M. T. and Oliveros, E., “Photochemical Technology”, John Wiley & Sons Inc., New York, U.S.A., 153 (1991).
Chen, J. Y., Wen, C. M. and Chen, T. L., “Effect of Oxygen Transfer on Lipase Production by Acinetobacter Radioresistens”,Biotechnol. Bioeng.,62(3), 311 (1999).
Cho, Y. S., Seo, K. W, Ahn, Y C. and Chung, Y S., “Preparation of BaO-PbO-Nd2O3-TiO2 Powders by Hydrothermal Synthesis”,Korean J. Chem. Eng.,19(4), 688 (2002).
Choi, Y. S. and Kim, B. W., “Photocatalytic Disinfection of Ecoli in a UV/TiO2-Immobilised Optical-Fiber Reactor”,J. Chem. Technol. Biotechnol.,75, 1145 (2000).
Davis, A. P., “Photocatalytic Oxidation of Organic Contaminants in Process Engineering for Pollution Control and Waste Minimization”, Marcel Dekker Inc., New York, U.S.A., 377 (1994).
Fernandez, A, Lassaletta, G., Jimenez, V. M., Justo, A., Gonzalez-Elipe, A. R., Hermann, J. M. and Tahiri, H., “Preparation and Characterization of TiO2 Photocatalysts Supported on Various Rigid Supports (Glass, Quartz and Stainless Steel): Comparative Studies of Photocatalytic Activity in Water Purification,”Appl. Catal.,B 7, 49 (1995).
Herrmann, J. M., Tahiri, H., Ait-Ichou, Y., Lassaletta, G, Gonzalez-Elipe, A. R. and Fernanez, A., “Characterization and Photocatalytic Activity in Aqueous Medium of TiO2 and Ag-TiO2 Coating on Quartz”,Appl. Catal.,B 13, 219 (1997).
Hoffmann, H. R., Martin, S. T, Choi, W. and Bahnemann, D. W., “Environmental Applications of Semiconductor Photocatalysis”,Chem. Rev.,95, 69 (1995).
Kuo, W S. and Lin, Y. T., “Photocatalytic Oxidation of Xenobiotics in Water with Immobilized TiO2 on Agitator”,J. Environ. Sci. Health,B 35(1), 61 (2000).
Lee, J. C., Kim, M. S. and Kim, B. W, “Removal of Paraquat Dissolved in a Photoreactor with TiO2 Immobilized on the Glass-Tube of UV Lamps”,Wat. Res.,36, 1776 (2002).
Legrini, O., Oliveros, E. and Braun, A. M., “Photochemical Processes for Water Treatment”,Chem. Rev.,93, 671 (1993).
Lindner, M., Theurich, J. and Bahnemann, D. W., “Photocatalytic Degradation of Organic Compounds: Accelerating the Process Efficiency”,Wat. Sci. Technol,35(4), 79 (1997).
Martyanov, N. and Savinov, E. N., “Mineralization of Organic Compounds in Photochemical and Photocatalytic Systems: Comparative Analysis for the Example of Methylviologen Photooxidation”,Catal. Today,39, 197 (1997).
Matsunaga, T. and Okochi, M., “TiO2-mediated Photochemical Disinfection ofEscherichia coli Using Optical Fibers”,Environ. Sci. Technol.,29, 501 (1995).
Matthews, R. W., “Photo-Oxidation of Organic Material in Aqueous Suspensions of Titanium Dioxide”,Water. Res.,20, 569 (1986).
Mazzarino, I., Piccinini, P. and Spinelli, L., “Degradation of Organic Pollutants in Water by Photochemical Reactors”,Catal. Today,48, 315 (1999).
Molinari, R., Mungari, M., Drioli, E., Paola, A. D. and Loddo, V., “Study on a Photocatalytic Membrane Reactor for Water Purification”,Catal. Today,55, 71 (2000).
Neves, J. M., Teixeira, J. A., Simoes, N. and Mota, M., “Effect of Airflow Rate on Yields of Steinernema Carpocapse Az 20 in Liquid Culture in an External-Loop Airlift Bioreactor”,Biotechnol. Bioeng.,72(3), 369 (2001).
Ollis, D. F., “Contaminant Degradation in Water”,Environ. Sci. Technol,19, 480 (1985).
Pope, J. D. and Benner, J. E., “Colorimetric Determination of Paraquat Residues in Soil and Water”,J. AOAC,57(1), 202 (1974).
Pozzo, R L., Baltanas, M. A. and Cassano, A. E., “Supported Titanium Dioxide as Photocatalyst in Water Decontamination: State of the ArÇ”Catal. Today,39, 219 (1997).
Shiraishi, F., Toyoda, K. and Fukinbara, S., “Photolytic and Photocatalytic Treatment of an Aqueous Solution Containing Microbial Cells and Organic Compounds in an Annular-Flow Reactor”,Chem. Eng. Sc,54, 1547 (1999).
Tennakone, K. I. and Kottegoda, R. M., “Photocatalytic Mineralization of Paraquat Dissolved in Water of TiO2 Supported on Polythene and Polypropylene Films”,J. Photochem. Photobiol.,A 93, 79 (1996).
Valladares, J. E., “A New Photocatalytic Reactor for the Photodegradation of Organic Contaminants in Water”, PhD. Thesis, Western Ontario University, Canada (1995).
Wu, Z. L., Yuan, Y I, Xing, Y. and Hu, Z. D., “Study on Volumetric Oxygen Transfer Coefficient of the Simulated Plant Cell Two-Phase Culture System”,Chem. Eng. Technol.,23(12), 1111 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, JC., Kim, MS., Kim, C.K. et al. Removal of paraquat in aqueous suspension of TiO2 in an immersed UV photoreactor. Korean J. Chem. Eng. 20, 862–868 (2003). https://doi.org/10.1007/BF02697289
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02697289