Summary
The aim of this work was to study the utilisation of coordinative europium compounds as stationary phase additives in gas chromatography.
The following series of aliphatic nucleophilic compounds were used as solutes: n-alcohols, carboxylic acid methyl esters, ketones, ethers and 1-alkenes.
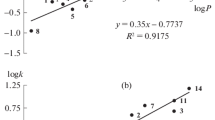
A correlation was established between the chromatographic retention values and the NMR chemical shift changes induced by interactions of the organic molecules with the europium chelate.
The agglomerate formation between the europium chelates and nucleophilic compounds involves changes in the NMR spectra and selective increases in the GC retention values when adding europium chelates to the stationary phases.
Similar content being viewed by others
References
I. Y. Slonim, A. H. Bulay, Usp. Khim.42, 1977 (1973).
R. Foster, C. A. Fyfe, Progr. NMR Spectrosc.4, 1 (1969).
J. C. Davis, K. H. Deb, Advances Magnetic Resonance Spectrosc.4, 201 (1970).
R. J. Laub, R. L. Pecsok, J. Chromatogr.114, 47 (1975).
V. A. Davankov, A. V. Semechkin, J. Chromatogr.141, 313 (1977).
V. Schurig, R. C. Chang, A. Zlatkis, B. Feibush, J. Chromatogr.99, 147 (1974).
B. Feibush, M. F. Richardson, R. E. Sievers, C. S. Springer, J. Amer. Chem. Soc.94, 6717 (1972).
J. E. Picker, R. E. Sievers, J. Chromatogr.203, 29 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kowalska, E.T., Kowalski, W.J. Gas chromatographic and NMR spectroscopic study of the interactions between the tris β-diketone chelates of europium and aliphatic nucleophiles. Chromatographia 19, 301–303 (1984). https://doi.org/10.1007/BF02687759
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02687759