Summary
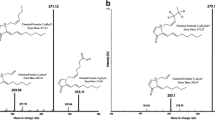
The synthetic prostaglandin F2α (PG2α), used as an ovulation regulator, and abortion inducer, is contaminated with its Δ5-trans isomer. The ratio of the isomers was measured by capillary GC.
The prostaglandin I2 (PGI2), a blood platelet aggregation inhibitor, easily decomposes to 6-ketoprostaglandin F1α (6-keto PG1α). Their ratio was measured by packed column GC.
An unexpected, instantaneous methanol cleavage was observed during the GC analysis of the methyl ketal derivative of 6-keto PG1α, forming a mixture of PGI2 and Δ6-prostaglandin I1 (Δ6-PGI1).
Similar content being viewed by others

References
Cs. Szántay, L. Novák, “Prosztaglandinok Szintézise” (Synthesis of Prostaglandins), Akadémiai Kiadó, Budapest, 1976.
R. A. Johnson, F. H. Lincoln, E. G. Nidy, W. P. Schneider, V. L. Thompson, U. Axen, J. Am. Chem. Soc.100, 7690 (1978).
Y. Chiang, A. J. Kresge, J. C. S. Chem. Comm. 1979, 129.
G. S. King, K. Blau, “Handbook of Derivatives from Chromatographyr Heyden & Son, London, 1977.
K. E. Rasmussen, J. Chromatogr.120, 491 (1976).
A. S. Christophersen, K. E. Rasmussen, J. Chromatogr.174, 454 (1979).
K. Grob Jr.,H. P. Neukom, J. High Resol. Chromatogr./Chromatogr. Commun.2, 15 (1979).
L. R. Snyder, J. Chromatogr. Sci.10, 200 (1972).
A. F. Pierce, “Silylation of Organic Compounds,” Pierce Chem. Co., Rockford, Illinois 1968.
V. Simonidesz, Zs. Gombos-Visky, G. Kovdes, E. Baitz-Gdcs, L. Radics, J. Am. Chem. Soc.100, 6756 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Juvancz, Z., Vajda, M. & Simonidesz, V. Gas chromatography of some prostaglandins. Chromatographia 19, 285–287 (1984). https://doi.org/10.1007/BF02687755
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02687755