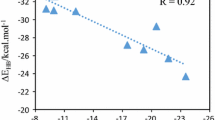
Abstract
The results of electronic structure calculations for o-benzoquinonediimine and its singly and doubly charged anions are presented. The variation of the geometrical and valence structures due to electron addition to the molecule is analyzed, and electron affinity is estimated. Polarizabilities of the neutral molecule and singly charged anion are calculated. The influence of the basis on the results of polarizability calculations for molecular anions is investigated.
Similar content being viewed by others
References
R. A. Metcalf and A. B. P. Lever, Inorg. Chem., 36, No. 21, 4762–4771 (1997).
P. R. Auburn and A. B. P. Lever, ibid., 29, No. 10, 2551–2553 (1990).
R. A. Metcalf, E. S. Dodsworth, S. S. Fielder, et al., ibid., 35, No. 26, 7741–7758 (1996).
H. Masui, A. B. P. Lever, and E. S. Dodsworth, ibid., 32, No. 3, 258–267 (1993).
Th. Jiistel, J. Bendix, N. Metzler-Nolte, et al., ibid., 37, No. 1, 24–35 (1998).
Th. H. Josefiak, J. E. Almlof, M. W. Feiereisen, and L. L. Miller, J. Am. Chem. Soc, 111, No. 11, 4105–4106 (1989); J. E. Almlöf, M. W. Feiereisen, Th. H. Josefiak, and L. L. Miller, ibid., 112, No. 3, 1206-1214 (1989).
M. W. Schmidt, K. K. Baldndge, J. A. Boatz, et al., J. Compui. Chem., 14, No. 11, 1347–1363 (1993).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., GA USSIAN 94, Revision D. 1, Gaussian Inc., Pittsburgh PA (1995).
A. D. Becke, J. Chem. Phys., 98, No. 11, 5648–5655 (1993).
C. Lee, W. Yang, and R. G. Parr, Phys. Rev. B, 37, No. 2, 785–793 (1988).
W. J. Hehre, R. F. Stewart, and J. A. Pople, J. Chem. Phys., 51, No. 7, 2657–2664 (1969).
W. J. Hehre, R. Dithfield, R. F. Stewart, and J. A. Pople, ibid., 52, No. 8, 2769–2773 (1970).
R. Dithfield, W. J. Hehre, and J. A. Pople, ibid., 54, No. 4, 724–728 (1971).
P. C. Hariharan and J. A. Pople, Theor. Chim. Acta, 28, No. 2, 213–222 (1973).
W. J. Hehre, R. Dithfield, and J. A. Pople, J. Chem. Phys., 56, No. 7, 2257–2261 (1972).
T. Clark, J. Chrandrasekhar, G. W. Spitznagel, and P. von R. Schleyer, J. Comput. Chem., 4, No. 3, 294–301 (1983).
C. B. Castellani and O. Cargo, Inorg. Chim. Acta, 150, No. 2, 119–124 (1988).
J. A. Foresman and A. Frisch, Exploring Chemistry with Electronic Structure Methods, Gaussian Inc., Pittsburgh PA (1993).
S. T. Howard, G. A. Attard, and H. F. Lieberman, Chem. Phys. Lett., 238, No. 1, 180–186 (1995).
P. W. Fowler and G. H. F. Diercksen, ibid, 167, Nos. 1/2, 105–110 (1990).
J. E. Greedy, G. B. Backskey, and N. S. Hush, Chem. Phys., 31, No. 2, 467 (1978).
P. W. Fowler and M. L. Klein, J. Chem. Phys., 65, No. 5, 3913 (1986).
T. Pluta, A. J. Sadlej, and R. J. Bartlett, Chem. Phys. Lett., 143, No. 1, 91–96 (1988).
G. H. F. Diercksen and A. J. Sadlej, Chem. Phys., 131, Nos. 215, 215–225 (1989).
T. Pluta, J. Noga, and R. J. Bartlett, Int. J. Quant. Chem. Quant. Chem. Symp., No. 28, 379-393 (1994).
S. A. Kucharski, Y. S. Lee, G. D. Purvis, and R. J. Bartlett, Phys. Rev. A, 29, No. 4, 1619–1626 (1984).
R. C. Weast (ed.). Handbook of Chemistry and Physics, CRC Press, Boca Raton, FL (1987).
A. N. Vereshchagin, Polarizability of Molecules [in Russian], Nauka, Moscow (1980).
Author information
Authors and Affiliations
Additional information
Translated from Zhumal Struktumoi Khim ii, Vol. 41, No. 1, pp. 34-41, January–February, 2000.
Rights and permissions
About this article
Cite this article
Baranovskii, V.I., Kuteikina-Teplyakova, A.V. & Panin, A.I. Electronic structure and polarizability of ortho-benzoquinonediimine and its singly and doubly charged anions. J Struct Chem 41, 28–34 (2000). https://doi.org/10.1007/BF02684724
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02684724