Abstract
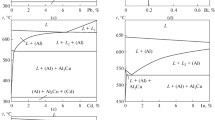
The activity of barium in liquid Al-Ba alloysX Ba ≤ 0.261) at 1373 K has been determined using the Knudsen effusion-mass loss technique. At higher concentrations (XBa ≥ 0.38), the activity of barium has been determined by the pseudo-isopiestic technique. Activity of aluminum has been derived by Gibbs-Duhem integration. The concentration-concentration structure factor of Bhatia and Thornton[22] at zero wave vector has been computed from the thermodynamic data. The behavior of the mean-square thermal fluctuation in composition indicates a tendency for association in the liquid state. The associated solution model with Al5Ba4 as the predominant complex has been used for the description of the thermodynamic behavior of liquid Al-Ba alloys. Thermodynamic data for the intermetallic compounds in the Al-Ba system and the enthalpy of mixing in liquid alloys have been derived using the phase diagram and the Gibbs’ energy of mixing of liquid alloys.
Similar content being viewed by others
References
M. Notin, B. Djamshidi, J.C1. Gachon, and J. Hertz:Thermochim. Acta, 1982, vol. 57, pp. 57–66.
B.P. Burylev, A.V. Vakhobov, and T.D. Dzhuraev:Russ. J. Phys. Chem., 1974, vol. 48 (6), pp. 809–11.
V.N. Vigdorovich, A.V. Vakhobov, and Yu.V. Plotnikov:Russ. J. Phys. Chem., 1972, vol. 46 (6), pp. 822–24.
R.P. Elliott and F.A. Shunk:Bull. Alloy Phase Diagrams, 1981, vol. 2 (3), pp. 351–53.
T.B. Massalski, J.L. Murray, L.H. Bennet, and H. Baker:Binary Alloy Phase Diagrams, ASM, Metals Park, OH, 1986, p. 92.
E. Albert:Z. Metallkd., 1934, vol. 26, pp. 6–9.
E.M. Flanigen: unpublished Master’s Thesis, Syracuse University, Syracuse, NY, 1952.
M. Iida:Nippon Kinzoku Gakkaishi, 1953, vol. 17, pp. 632–34.
G. Bruzzone and F. Merlo:J. Less-Common Met., 1975, vol. 39, pp. 1–6.
Hultgren, P.D. Desai, D.T. Hawkins, M. Gleiser, and K.K. Kelley:Selected Values of the Thermodynamic Properties of the Elements, ASM, Metals Park, OH, 1973, p. 59.
M.L. Fornasini and G. Bruzzone:J. Less-Common Met., 1975, vol. 40, pp. 335–40.
M.L. Fornasini:Acta Crystallogr., 1975, vol. B31, pp. 2551–52.
V.M. Nepochatov, E.V. Kiseleva, and A.A. Bundel:Russ. J. Phys. Chem., 1979, vol. 53 (4), pp. 588–89.
S.T. Kulichikina and B.P. Nikonov:Russ. J. Phys. Chem., 1967, vol. 41 (9), pp. 1167–69.
Ya. L. Kharif, P.V. Kovtunenko, A.A. Maier, I.Kh. Avetisov, and M.M. Bebakin:Inorg. Mater., 1984, vol. 20, pp. 1182–86.
K.T. Jacob:Z. Metallkd., 1985, vol. 76, pp. 415–19.
K.T. Jacob, S. Srikanth, and Y. Waseda:Trans. Jpn. Inst. Met., 1988, vol. 29, pp. 50–59.
S. Srikanth and K.T. Jacob: Z.Metallkd., 1991, in press.
R.P. Iczkowski, J.L. Margrave, and S.M. Robinson:J. Phys. Chem., 1963, vol. 67, pp. 229–33.
K.T. Jacob and Y. Waseda:J. Less-Common Met., 1988, vol. 139, pp. 249–59.
J.H. Norman and P. Winchell: inPhysico-Chemical Measurements in Metals Research, R.A. Rapp, ed., Interscience Publishers, New York, NY, 1970, vol. IV.
A.B. Bhatia and D.E. Thornton:Phys. Rev., 1970, vol. 2B, pp. 3004–12.
A.S. Jordan:Metall. Trans., 1970, vol. 1, pp. 239–49.
S. Srikanth and K.T. Jacob:Metall. Trans. B, 1988, vol. 19B, pp. 465–70.
S. Srikanth and K.T. Jacob:CALPHAD, 1989, vol. 13, pp. 149–58.
P.J. Flory:J. Chem. Phys., 1942, vol. 10, pp. 51–61.
P.J. Flory:Principles of Polymer Chemistry, Cornell University Press, Ithaca, NY, 1953.
M.J.D. Powell:Comput. J., 1964, vol. 7, pp. 155–62.
E.A. Brandes:Smithell’s Metals Reference Book, 6th ed., Butterworth’s, London, 1983, p. 14–7.
Author information
Authors and Affiliations
Additional information
Formerly with the Department of Metallurgy, Indian Institute of Science.
Rights and permissions
About this article
Cite this article
Srikanth, S., Jacob, K.T. Thermodynamics of aluminum-barium alloys. Metall Trans B 22, 607–616 (1991). https://doi.org/10.1007/BF02679015
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02679015