Abstract
Karádi O, Bódis B, Király Á, Abdel-Salam OME, Sütō G, Vincze Á, Mózsik Gy. Surgical vagotomy enhances the indomethacin-induced gastrointestinal mucosal damage in rats. Inflammopharmacology. 1994;2:389-399.
Indomethacin (IND)-induced gastrointestinal mucosal lesions and changes of vascular permeability were studied in rats with and without acute bilateral surgical vagotomy.
The aims of study were (a) to compare IND-induced mucosal lesions in the stomach, small intestine and large bowel of rats with intact vagus and acute surgical vagotomy and (b) to evaluate the changes of vascular permeability following these treatments.
The gastrointestinal (GI) mucosal damage was produced by IND (20 mg/kg s.c.) at 24 and 48 h after IND administration. Sham operation (laparotomy alone) or surgical vagotomy alone was carried out in control animals. The number and severity of GI mucosal damage was determined. The changes of mucosal vascular permeability were determined by Evans Blue assays in the GI mucosa and in the contents of stomach, small intestine and colon. Evans Blue was given into the tail vein at 15 min before the killing of animals.
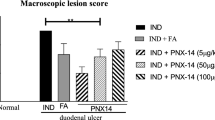
It was found that (a) no ulceration and change in mucosal vascular permeability was found in any parts of GI tract after sham operation or surgical vagotomy (without IND); (b) IND caused mucosal injury coincidental with vascular permeability in the stomach and small intestine, but not in the large bowel; (c) surgical vagotomy aggravated the IND-induced mucosal damage and increased vascular permeability in the stomach and small intestine, but not in the colon. In conclusion, the intact vagal nerve is required for the prevention of gastric and small intestinal mucosa to protect against IND injury.
Similar content being viewed by others
References
Djahanguiri B. The production of acute gastric ulceration by indomethacin in the rat. Scand J Gastroenterol. 1969;4:265–7.
Brodie DA, Cook PG, Bauer BJ. Indomethacin-induced intestinal lesions in the rat. Toxicol Appl Pharmacol. 1970;17:615–24.
Szabo S, Mózsik Gy, eds. New pharmacology of ulcer disease: experimental and new therapeutic approaches. New York: Elsevier Publishing Company. 1987.
Szabo S, Trier JS, Brown A, Schnoor J. Early vascular injury and increased permeability in gastric mucosal injury caused by ethanol in the rat. Gastroenterology. 1985;88:228–32.
Miller TA. Protective effects of prostaglandins against gastric and mucosal damage: current knowledge of proposed mechanisms. Am J Physiol. 1983;245:6601–5.
Mózsik Gy, Moron F, Jávor T. Cellular mechanisms of the development of gastric mucosal damage and gastric cytoprotection induced by prostacyclin on rats. A pharmacological study. Prostaglandin LeukMed. 1982;9:71–84.
Mózsik Gy, Király Á, Garamszegi M, Jávor T, Nagy L, Németh A, Sütō G, Vincze Á. Mechanisms of vagal nerve in gastric mucosal defense: unchanged gastric emptying and increased vascular permeability. J Clin Gastroenterol. 1992;14(Suppl1):S140–4.
Mózsik Gy, Király Á, Garamszegi M, Jávor T, Nagy L, Németh A, Sütō G, Vincze Á. Gastric cytoprotection mediating in SH-groups is failured by surgical vagotomy. Acta Physiol Hung. 1990;75(Suppl 1):219–20.
Mózsik Gy, Karádi O, Király Á, Matus Z, Sütō G, Tóth Gy, Vincze Á. Vagal nerve and the gastric mucosal defense. J Physiol. 1993;87:59–64.
Mózsik Gy, Király Á, Garamszegi M, Jávor T, Nagy L, Sütō G, Tóth Gy, Vincze Á. Failure of prostacycline, β-carotene, atropine and cimetidine to produce gastric cyto- and general mucosal protection in surgically vagotomized rats. Life Sci. 1991;49:1383–9.
Sütō G, Király Á, Vincze Á, Jávor T, Mózsik Gy,A new computer assisted method to determine the area of mucosal lesions in the stomach induced by chemicals in rats. Z Gastroenterol. 1991;29(4):205–6.
Rainsford KD, Brune K. Selective cytotoxic actions of aspirin on parietal cells: a principal factor in the early stages of aspirin-induced gastric damage. Arch Toxicol. 1978;40:143–50.
Rainsford KD, Fox SA, Osborne DJ. Relationship between drug absorption, inhibition of cyclooxygenase and lipoxygenase pathways and the development of gastric mucosal damage by non-steroidal anti-inflammatory drugs in rats and pigs. In: Bailey MJ, ed. Advances in prostaglandins, leukotrienes and lipoxins. New York: Plenum Press; 1985:639–53.
McCormack K, Brune K. Classical absorption theory and the development of gastric mucosal damage associated with the non-steroidal anti-inflammatory drugs. Arch Toxicol. 1987;60:261–9.
McCormack K. Mathematical model for assessing risk of gastrointestinal reactions to NSAIDs. In: Rainsford KD, ed. Azapropazone: over two decades of clinical use, 2nd edn. Dordrecht: Kluwer Academic Publishers; 1991:81–94.
Mózsik Gy, Király Á, Garamszegi M, Nagy L, Sütō G, Tóth Gy, Vincze Á, Jávor T. Vagus in gastric mucosal injury and prevention. Dig Dis Sci. 1989;34:1319.
Grijalva CV, Novin D. The role of the hypothalamus and dorsal vagal complex in gastrointestinal function and pathophysiology. Ann NY Acad Sci. 1990;597:207–22.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Karádi, O., Bödis, B., Király, Á. et al. Surgical vagotomy enhances the Indomethacin–Induced gastrointestinal mucosal damage in rats. Inflammopharmacology 2, 389–399 (1994). https://doi.org/10.1007/BF02678605
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02678605