Abstract
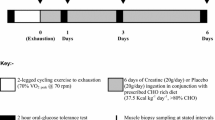
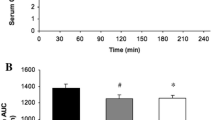
The rate of glycogen resynthesis in human skeletal muscle after glycogen-depleting exercise is known to depend on carbohydrate intake and is reported to reach a platean after an adequate amount of carbohydrate (CHO) consumption. Efforts to maximize the rate of glycogen storage by changing the type and form of CHO, as well as by adding proteins or lipids have yielded inconsistent results. The objective of this study was to assess whether isocaloric addition of proteins and arginine to a CHO diet in the first 4 h after an endurance exercise would increase the rate of glycogen synthesis. The CHO solution, given twice at a 2 h interval according to earlier optimized protocols, contained 1.7 g CHO kgbody weight. The effects of this solution were compared to those of an isocaloric solution containing 1.2 g CHO/kgbody weight plus 0.5 g protein/kgbody weight (including 5 g arginine). Glycogen was measured in quadriceps muscle in vivo with natural abundance13C-magnetic resonance spectroscopy before exercise and twice after exercise, before and at the end of a 4-h period following the intake of one of the solutions. Eight subjects took part in a randomized cross-over trial separated by at least 1 week. Glycogen synthesis was found to be significantly increased with both regimes compared to a zero-caloric placebo diet, but no significant difference in glycogen resynthesis was found between the CHO-only diet and the one supplemented by proteins and arginine. It is estimated that significance would have been reached for an increase of 34%, while the effectively measured synthesis rates only differed by 5%.
Similar content being viewed by others
References
Bergstrom J, Hermansen L, Hultman E, Saltin B. Diet. muscle glycogen and physical performance. Acta Physiol Scand 1967;71:140–50.
Ivy JL, Katz AL, Cutler CL, Sherman WM, Coyle EF. Muscle glycogen synthesis after exercise: effect of time of carbohydrate ingestion. J Appl Physiol 1988;64:1480–5.
Ivy JL, Lee MC, Brozinick JT, Jr., Reed MJ. Muscle glycogen storage after different amounts of carbohydrate ingestion. J Appl Physiol 1988;65:2018–23.
Bergstrom J, Hultman E. Muscle glycogen synthesis after exercise: an enhancing factor localized to the muscle cells in man. Nature 1966;210:309–10.
Blom PC, Hostmark AT, Vaage O, Kardel KR, Maehlum S. Effect of different post-exercise sugar diets on the rate of muscle glycogen synthesis. Med Sci Sports Exerc 1987;19:491–6.
Reed MJ, Brozinick JT, Jr., Lee MC, Ivy JL. Muscle glycogen storage postexercise: effect of mode of earbohydrate administration. J Appl Physiol 1989;66:720–6.
Keizer HA, Kuipers H, van Kranenburg G, Geurten P. Influence of liquid and solid meals on muscle glycogen resynthesis, plasma fuel hormone response, and maximal physical working capacity. Int J Sports Med 1987;8:99–104.
Burke LM, Collier GR, Hargreaves M. Muscle glycogen storage after prolonged exercise: effect of the glycemic index of carbohydrate feedings. J Appl Physiol 1993;75:1019–23.
Zawadzki KM, Yaspelkis BB, III, Ivy JL. Carbohydrate-protein complex increases the rate of muscle glycogen storage after exercise. J Appl Physiol 1992;72:1854–9.
Tarnopolsky MA, Bosman M, Macdonald JR, Vandeputte D, Martin J, Roy BD. Postexercise protein-carbohydrate and carbohydrate supplements increase muscle glycogen in men and women. J Appl Physiol 1997;83:1877–83.
Floyd JC, Jr., Fajans SS, Conn JW, Knopf RF, Rull J. Insulin secretion in response to protein ingestion. J Clin Invest 1966;45:1479–86.
Floyd JC, Jr., Fajans SS, Conn JW, Knopf RF, Rull J. Stimulation of insulin secretion by amino acids. J Clin Invest 1966;45:1487–502.
Floyd JC, Jr., Fajans SS, Pek S, Thiffault CA, Knopf RF, Conn JW. Synergistic effect of essential amino acids and glucose upon insulin secretion in man. Diabetes 1970;19:109–15.
Danforth WH, Harvey P. Glycogen synthetase and control of glycogen synthesis in muscle. Biochem Biophys Res Commun 1964;16:466–71.
Cohen P, Nimmo HG, Proud CG. How does insulin stimulate glycogen synthesis? Biochem Soc Symp 1978;43:69–95.
Balon TW, Nadler JL. Evidence that nitric oxide increases glucose transport in skeletal muscle. J Appl Physiol 1997;82:359–63.
Giugliano D, Marfella R, Verrazzo G, Acampora R, Coppola L, Cozzolino D, D’Onofrio F. The vascular effects ofl-arginine in humans. The role of endogenous insulin. J Clin Invest 1997;99:433–8.
Avison MJ, Rothman DL, Nadel E, Shulman RG. Detection of human muscle glycogen by natural abundance 13C NMR. Proc Natl Acad Sci USA 1988;85:1634–6.
Jue T, Rothman DL, Tavitian BA, Shulman RG. Natural-abundance C-13 NMR study of glycogen repletion in human liver and muscle. Proc Natl Acad Sci USA 1989;86(5):1439–42.
Beckmann N, Seelig J, Wick H. Analysis of glycogen storage disease by in vivo 13C NMR: Comparison of normal volunteers with a patient. Magn Reson Med 1990;16:150–60.
Jehenson P, Duboc D, Bloch G, Fardeau M, Syrota A. Diagnosis of muscular glycogenosis by in vivo natural abundance 13C NMR spectroscopy. Neuromuscul Disord 1991;1:99–101.
Taylor R, Price TB, Katz LD, Shulman RG, Shulman GI. Direct measurement of change in muscle glycogen concentration after a mixed meal in normal subjects. Am J Physiol 1993;265:E224–9.
van den Bergh AJ, Houtman S, Heerschap A, Rehrer NJ, van den Boogert HJ, Oeseburg B, Hopman MT. Muscle glycogen recovery after exercise during glucose and fructose intake monitored by 13C-NMR. J Appl Physiol 1996;81:1495–500.
Taylor R, Price TB, Rothman DL, Shulman RG, Shulman GI. Validation of 13C NMR measurement of human skeletal muscle glycogen by direct biochemical assay of needle biopsy samples. Magn Reson Med 1992;27:13–20.
Slein MW. In: Bergmeyer HW. editor. Methods of Enzymatic Analysis. Weinheim: Verlag Chemie Academic Press, 1965:117.
Herbert V, Lau KS, Gottlieb CW, Bleicher SJ. Coated charcoal immunoassay of insulin. J Clin Endocrinol Metab 1965;25:1375–84.
Issekutz BJ, Rodahl K. Respiratory quotient during exercise. J Appl Physiol 1961;16:606–10.
Howley ET, Bassett DR, Jr., Welch HG. Criteria for maximal oxygen uptake: review and commentary. Med Sci Sports Exerc 1995;27:1292–301.
Choi D, Cole KJ, Goodpaster BH, Fink WJ, Costill DL. Effect of passive and active recovery on the resynthesis of muscle glycogen. Med Sci Sports Exerc 1994;26:992–6.
Silver MS, Joseph RI, Hoult DI. Highly selective Pi,2 and Pi pulse generation. J Magn Reson 1984;59:347–51.
Van Cauteren M, Miot F, Segebarth CM, Eisendrath H, Osteaux M, Willem R. Excitation characteristics of adiabatic halfpassage RF pulses used in surface coil MR spectroscopy. Application to 13C detection of glycogen in the rat liver. Phys Med Biol 1992;37:1055–64.
Shellock FG, Schaefer DJ, Kanal E. Physiologic responses to an MR imaging procedure performed at a specific absorption rate of 6.0 W kg. Radiology 1994;192:865–8.
Slotboom J, Boesch C, Kreis R. Versatile frequency domain fitting using time domain models and prior knowledge. Magn Reson Med 1998;39:899–911.
Overloop K, Van Hecke P, Vanstapel F, Chen H, van Huffel S, Knijn A, van Ormondt D. Evaluation of signal processing methods for the quantification of a multi-exponential signal: the glycogen 13C-1 NMR signal. NMR Biomed 1996;9:315–21.
Kreis R, Koster M, Kamber M, Hoppeler H, Boesch C. Peak assignment in localized IH MR spectra based on oral creatine supplementation. Magn Reson Med 1997;37:159–63.
Kreis R, Jung B, Slotboom J, Felblinger J, Boesch C. Effect of exercise on the creatine resonances in IH-MR spectra of human skeletal muscle. J Magn Reson 1999;137:350–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rotman, S., Slotboom, J., Kreis, R. et al. Muscle glycogen recovery after exercise measured by13C-magnetic resonance spectroscopy in humans: effect of nutritional solutions. MAGMA 11, 114–121 (2000). https://doi.org/10.1007/BF02678474
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02678474