Abstract
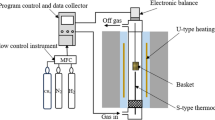
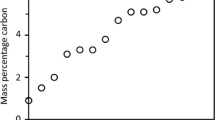
The rates of carburization by mixtures of CO, CO2, H2 and H2O in N2 have been measured gravimetrically at 925 °. The rate equation which best describes the experimental data is based on a mechanism which involves a rapid surface dissociation of CO into carbon and oxygen atoms, and a subsequent rate determining step between this atomic oxygen and either CO or H2. The CO-H2 system carburizes much faster than CO alone, because H2 combines faster with atomic oxygen than does CO. The carburizing rate constant for CO-H2 is 44 times that for CO alone. The mechanism is confirmed by the additivity of the separate rates for the CO-H2 mixtures and for CO alone.
Similar content being viewed by others
References
R. J. Fruehan and L. J. Martonik:High Temp. Sci., 1971, vol. 3, pp. 244–56.
R. J. Fruehan:Met. Trans., 1973, vol. 4, pp. 2123–27.
H. J. Grabke:High Temperature Gas-Metal Reactions in Mixed Environments, J. A. Jansson and Z. A. Foroulis, eds., pp. 130–42, AIME, 1973.
H. J. Grabke:Arch. Eisenhuettenwes., 1975, vol. 46, p. 75; Brutcher translation HB 9633.
S. R. Shatynski and H. J. Grabke:Arch. Eisenhuettenwes., 1978, vol. 49, pp. 129–33.
E. T. Turkdogan and P. Grieveson:J. Electrochem. Soc, 1967, vol. 114, pp. 59–64.
K. J. Best and H. J. Grabke:Ber. Bunsenges. Phys. Chem., 1971, vol. 75, pp. 524–32.
C. Wells, W. Batz, and R. F. Mehl:Trans. AIME, 1950, vol. 188, pp. 553–60.
R. W. Gurry:Trans. AIME, 1942, vol. 150, pp. 172–84.
T. Ellis, I. M. Davidson, and C. Bodsworth:J. Iron Steel Inst., 1963, vol. 201, pp. 582–87.
U.K. Kishi and M. W. Roberts:J. Chem. Soc. Faraday Trans., 1975, vol. 71, pp. 1715–20.
“JANAF Thermochemical Tables,” 2nd ed., National Bureau of Standards, 1971.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kaspersma, J.H., Shay, R.H. Carburization of iron by CO-based mixtures in nitrogen at 925 °C. Metall Trans B 12, 77–83 (1981). https://doi.org/10.1007/BF02674760
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02674760