Abstract
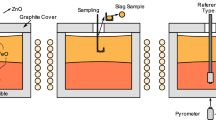
A novel reaction scheme for transforming certain metal sulfides to the corresponding oxides has been developed. In this process, steam oxidizes the sulfide into the oxide, and the hydrogen sulfide produced reacts with lime to form calcium sulfide and regenerate steam. There is no net consumption or generation of gaseous species. Thus, the overall reaction can be carried out in a closed system as far as the gas phase is concerned. This eliminates the possibility of emitting hydrogen sulfide out of the reactor. Only certain metal sulfides are thermodynamically amenable to this treatment. In this paper, the reaction of ZnS to ZnO by this scheme is described, together with a detailed formulation of the rate equation for the overall reaction based on the kinetics of the component gas-solid reactions. Although the present work was done with CaO, other suitable oxides may be used in its place. A further potential application of this process is to the selective oxidation of certain sulfide(s) from complex sulfide ores as a treatment prior to the separation of minerals.
Similar content being viewed by others
Abbreviations
- A :
-
gaseous speciesA (= H2O in this work)
- b :
-
number of moles ofB reacted by one mole ofA (=1 in this work)
- B :
-
solid reactantB (=ZnS in this work)
- C :
-
gaseous speciesC (=H2S in this work)
- C i :
-
molar concentration of speciesi
- C T :
-
sum of molar concentrations ofA andC
- d :
-
number of moles ofD reacted by one mole ofC (=1 in this work)
- D :
-
solid reactantB (=CaO in this work)
- f(X B ):
-
conversion function defined in Eq. [11]
- g(X D ):
-
conversion function defined in Eq. [12]
- k 1,k 2 :
-
reaction-rate constants for reactions [1] and [2], respectively
- K 1,K 2 :
-
equilibrium constants for reactions [1] and [2], respectively
- n :
-
constant in Eq. [11]
- n i :
-
number of moles of speciesi
- t :
-
time
- v 1,v 2 :
-
net forward rates of reactions [4] and [5]
- x i :
-
fractional conversion of solid speciesi
- αi :
-
fraction of total solid volume occupied by speciesi
- γ:
-
molar ratio defined by Eq. [21]
- λ:
-
pore-blocking rate constant in Eq. [12]
- ρi :
-
true molar density of solid speciesi
References
F. P. Haver and M. M. Wong:Mining Eng., 1972, vol. 24, no. 6, pp. 52–53.
R. W. Bartlett and H. H. Haung:J. Metals, 1973, vol. 25, no. 12, pp. 28–34.
H. Y. Sohn: “Process for Treating Sulfide-Bearing Ores,” U.S. Patent No. 4,376,647, March 15, 1983.
H. Y. Sohn and D. Kim:J. Metals, 1984, vol. 36, no. 1, pp. 67–73.
D. Kim: Ph.D. Dissertation, University of Utah, Salt Lake City, UT, 1980.
J. F. Elliott and M. Gleiser:Thermochemistry of Steel Making, Addison-Wesley, London, 1960, vol. 1.
T. B. Reed:Free Energy of Formation of Binary Compounds, The MIT Press, 1971.
H. Y. Sohn and D. Kim:Metall. Trans. B, 1987, vol. 18B, pp. 451–57.
H. Y. Sohn and J. Szekely:Chem. Eng. Sci., 1973, vol. 28, pp. 1789–801.
S. Won and H. Y. Sohn:Metall. Trans. B, 1985, vol. 16B, pp. 163–68.
F. Habashi and B. I. Yostos:J. Metals, 1977, vol. 29, no. 7, pp. 11–16.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sohn, H.Y., Kim, D. Kinetics of zinc oxide formation from zinc sulfide by reaction with lime in the presence of water vapor. Metall Trans B 18, 727–732 (1987). https://doi.org/10.1007/BF02672890
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02672890