Abstract
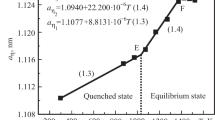
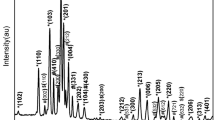
The Cr-Ni-V system was investigated at 1100 ‡C using XRD, metallography, and energy dispersive chemical analysis of phases using scanning electron microscopy (SEM). The bcc α phase extends from the Cr-Ni binary to the V-Ni binary and has a maximum solubility of Ni in ternary α phase alloys of ∼12 at. %. A wide fcc γ phase field exists at the Ni corner. A ternary Σ phase (Σ2) exists; it differs from the binary Ni-V Σ phase (Σ1). The Σ1 phase has a tetragonal unit cell with parameters almost double those of a normal Σ phase (Σ2). Results indicate difficulty in achieving equilibrium states in alloys containing more than 30 at. % V and less than 15 at. % Cr even after 480 h annealing. The phase analysis of alloys annealed for 170 to 480 h suggests Σ2 first forms through a ternary peritectic reaction to subsequently develop the Σ1, phase by subsidiary reactions in those ternary alloys in the concentration range where Σ1 is the stable phase. A tentative reaction scheme, consistent with the phase relations established at 1100 ‡C, is proposed.
Similar content being viewed by others
Cited References
W.B. Pearson and W. Hume-Rothery, “The Constitution and Structure ofNi-V Alloys in the Region 0–50 at. pct. V,≓J. Inst. Met., 80, 641–652(1952).
A.A. Kodentzov, S.F. Dunaev, and E.M. Slusarenko, “Determination of the Phase Diagram of the V-Ni-Cr System Using Diffusion Couples and Equilibrated Alloys,≓J. Less-Common Met., 135, 15–24 (1987).
A.K. Malhotra and K.P. Gupta, “Phase Equilibria in Cr-Ni-V System at 1100‡C≓J. Alloy Phase Diagrams, 5, 132–135 (1989).
K.P. Gupta, “Phase Diagrams of Ternary Nickel Alloys, Part 1,≓ Indian Institute of Metals, Calcultta, India, 1990.
K.P. Gupta, “Phase Diagrams of Ternary Nickel Alloys, Part 2,≓ Indian Institute of Metals, Calcultta, India, 1991.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Singh, S.K., Gupta, K.P. The Cr-Ni-V system. JPE 16, 129–136 (1995). https://doi.org/10.1007/BF02664850
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02664850