Abstract
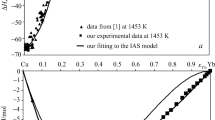
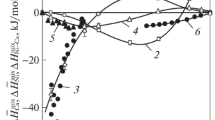
The activities of Cu and CuO0.5 were measured in melts in the systems Cu-O, Cu-O-SiO2, and Cu-O-CaO at 1300 °C by establishing the equilibrium Cu(1) + l/4O2(g) = CuO0.5(l) between the melts, atmospheric air, and a small piece of platinum wire immersed in the melt. Chemical analysis of the equilibrium Pt-Cu alloy which was formed, with knowledge of the activity-composition relationship in the system Pt-Cu, allowed determination of the activity of copper in the melt. The activity of CuO0.5 along the air isobar in the system Cu-O-SiO2 decreases from unity to 0.64 at the solubility limit ofX SiO 2 = 0.13 and, in the system Cu-O-CaO, decreases to 0.7 at the solubility limit ofX CaO = 0.34. The activity of CuO0.5 is well represented by the regular solution model over the limited range of liquid composition in the system Cu-O-SiO2 and is reasonably well represented by the model in melts in the system Cu-O-CaO.
Similar content being viewed by others
References
A. Luraschi and J.F. Elliott:Extractive Metallurgy of Copper, J.C. Yannopoulos and J.C. Agarwal, eds., TMS, Warrendale, PA, 1976, vol. 1, pp. 90–114.
U. Kuxmann and K. Kune:Erzmetall., 1968, vol. 21, p. 199.
C. Landolt: Ph.D. Thesis, Pennsylvania State University, University Park, PA, 1969.
C. Diaz:The Thermodynamic Properties of Copper-Slag Systems, International Copper Research Association, New York, NY, 1974, pp. 145–49.
Y.-G. Park and D.R. Gaskell:Metall. Trans. B, 1989, vol. 20B, pp. 127–35.
R. Schmid:Metall. Trans. B, 1983, vol. 14B, pp. 473–81.
S.G. Whiteway, I.B. Smith, and C.R. Masson:Can. J. Chem., 1970, vol. 48, pp. 33–45.
R. Altmann:Trans. IMM, Sect. C, 1978, vol. 87, pp. C23-C28.
Author information
Authors and Affiliations
Additional information
Formerly Graduate Student, University of Illinois, Urbana-Champaign, IL
Rights and permissions
About this article
Cite this article
Peddada, S.R., Gaskell, D.R. The activity of CuO0.5 along the air isobars in the systems Cu-O-SiO2 and Cu-O-CaO at 1300 °C. Metall Trans B 24, 59–62 (1993). https://doi.org/10.1007/BF02657872
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02657872