Abstract
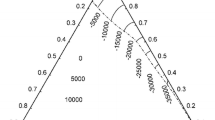
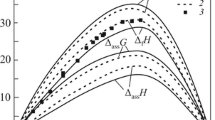
By means of high temperature calorimetry the mixing enthalpies ΔH of liquid Li-Sn alloys have been measured; however, due to experimental problems they were determined only forx Li = 0.01 to 0.5 andx Li = 0.87 to 0.99. The range of temperatures studied was 691 to 938 K. High compound forming tendency in Li-Sn is reflected by a triangular-shaped relation for ΔH vs x Li. The extrapolated maximum of this plot is about −40 kJ mol−1 close to Li4Sn. Using the concentration cell Bi(l)Li3Bi(s)¦ LiF-LiCl¦Li-Sn(l) the emf was measured as function of temperature (775 to 906 K) atx Li = 0.1 to 0.603 enabling calculations of partial thermodynamic data for lithium in liquid Li-Sn solutions. Integral enthalpies calculated from partial enthalpies of lithium correspond well to the calorimetrically obtained integral mixing enthalpies in the concentration range where both emf and calorimetric data were obtained. The extrapolated maximum of ΔH from calorimetric studies and minimum of integral excess entropies from emf measurements correlate well with results of structure measurements and of other structure sensitive properties. All this experimental information indicates a maximum chemical short range order close to the composition Li4Sn.
Similar content being viewed by others
References
F. Sommer:Z. Metallkde., 1982, vol. 73, pp. 72–86.
F. Sommer:Ber. Bunsenges. Phys. Chem., 1983, vol. 87, pp. 749–56.
Z. Moser and R. Castanet:Metall. Trans. B, 1979, vol. 1OB, pp. 483–81.
R. Castanet, Z. Moser, and W. Gasior:CALPHAD, 1980, vol. 4, pp. 234–40.
Z. Moser, E. Kawecka, F. Sommer, and B. Predel:Metall. Trans. B, 1982, vol. 13B, pp. 71–76.
J. F. Smith and Z. Moser:Journal of Nuclear Materials, 1976, vol. 5, pp. 158–74.
M.S. Foster, C.E. Crouthamel, and S.E. Wood:J. Phys. Chem., 1966, vol. 70, pp. 3042–45.
A. G. Morachevski, L. N. Gerasimenko, A.I. Demidov, and O.A. Drozdoba:Elektrohimya, 1972, vol. 8, pp. 1662–24.
M.L. Saboungi and M. Blander:J. Electrochem. Soc, 1977, vol. 124, pp. 6–13.
C. J. Wen and R. A. Huggins:J. Electrochem. Soc, 1981, vol. 128, pp. 1181–87.
A. K. Fischer and S. A. Johnson:J. Chem. Eng. Data, 1972, vol. 17, pp. 280–83.
P. Baradel, A. Vermande, I. Ansara, and P. Desré:Rev. Int. Hautes Temper, et Refract., 1971, vol. 8, pp. 201–04.
B. Predel, G. Oehme, and W. Vogelbein:Z. Metallkde., 1978, vol. 69, pp. 163–69.
W. Becker, G. Schwitzgebel, and H. Ruppersberg:Z. Metallkde., 1981, vol. 72, pp. 186–90.
A.D. Pelton and S.N. Flengas:Can. J. Chem., 1969, vol. 47, p. 2283.
F. Sommer, J.J. Lee, and B. Predel:Z. Metallkde., 1980, vol. 71, pp. 818–21.
G. Oehme and B. Predel:Thermodynamic Acta, 1978, vol. 23, pp. 267–73.
B. P. Alblas, W. van der Lugt, J. Dijkstra, and C. van Dijk:J. Phys. F: Met. Phys., 1984, vol. 14, pp. 1995–2005.
C.van der Marel, W. Geertsma, and W. van der Lugt: J. Phys. F:Metal Phys., 1980, vol. 10, p. 2305.
C. van der Marel, A. B. van Oosten, A. B. Geertsma, and W. van der Lugt:J. Phys. F: Met. Phys., 1982, vol. 12, pp. 2349–61.
M. Hansen and K. Anderko:Constitution of Binary Alloys, McGraw-Hill, New York, NY, 1958.
W. Müller:Z. Naturforschg., 1974, vol. 29b, p. 304.
V. Frank and W. Müller:Z. Naturforschg., 1975, vol. 30b, p. 316.
D. M. Bailey, W. H. Skelton, and J. F. Smith:J. Less-Common Met., 1979, vol. 64, pp. 233–40.
M.L. Sabounghi, J. Marr, and M. Blander:J. Chem. Phys., 1978, vol. 68, p. 1375.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moser, Z., Gasior, W., Sommer, F. et al. Calorimetric and emf studies on liquid Li-Sn alloys. Metall Trans B 17, 791–796 (1986). https://doi.org/10.1007/BF02657142
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02657142