Abstract
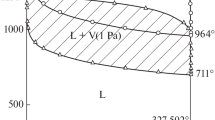
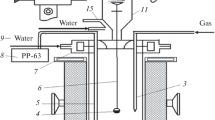
The kinetics of vaporization of lead sulfide were investigated in the temperature range 785 to 993 °C using a thermogravimetric balance. The effect of nonreactive ambient atmosphere on the vaporization kinetics was examined in some detail. Under He-rich He-N2 atmosphere the rate of vaporization was found to be about four times as large, as that under N2-atmosphere. A mathematical model was developed taking into consideration the chemical kinetic and mass-transfer factors. It was found that faster vaporization rates obtained with He-rich atmosphere are due to the higher diffusivity of PbS(g) in He(g). The chemical kinetic aspects of vaporization appear to be unaffected by the ambient atmosphere. The activation energy for vaporization has a value of 198.95 kJ·mole-1 in the temperature range investigated. The evaporation coefficient was found to have values ranging from 0.05 to 0.14.
Similar content being viewed by others
References
E.R. Cole, Jr., W.M. Dressel, and M.M. Fine: Report of Investigation 7926, Bureau of Mines, U.S. Department of the Interior, Washington, DC, 1974.
G.A. Somorjai: inCondensation and Evaporation of Solids, E. Rutner, P. Goldfinger, and J.P. Hirth, eds., Gordon and Breach, New York, NY, 1964, p. 417.
C.T. Ewing and K. H. Stern:J. Phys. Chem., 1973, vol. 77, p. 1442.
C.T. Ewing, and K.H. Stern:J. Phys. Chem., 1975, vol. 79, p. 2007.
D.L. Howleh, J.E. Lester, and G.A. SomorjaiJ. Phys. Chem., 1971, vol. 75, p. 4049.
J.C. Halle, and K.H. Stern:J. Phys. Chem., 1980, vol. 84, p. 1699.
S.T. Lam, and Z.A. Munir:J. Crystal. Growth, 1981, vol. 51, p. 227.
J.E. Lester, and G.A. Somorjai:J. Chem. Phys., 1968, vol. 49, pp. 2940–48.
G.M. Rothberg, M. Eisenstadt, and P. Kusch:J. Chem. Phys., 1959, vol. 30, pp. 517–27.
P.B. Weisz, and A.B. Schwartz:J. Catal 1962, vol. 1, p. 399.
R.B. Bird, W.E. Stewart, and E.N. Lightfoot:Transport Phenomena, John Wiley & Sons, New York, NY, 1960, p. 570.
O. Kubaschewski, and C.B. Alcock:Metallurgical Thermochemistry, 5th edn., Pergamon Press, New York, NY, 1979, p. 368.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
El-Rahaiby, S.K., Rao, Y.K. Kinetics of vaporization of lead sulfide. Metall Trans B 13, 633–641 (1982). https://doi.org/10.1007/BF02650021
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02650021