Abstract
Thermodynamics have long been applied to our understanding of the reactive wetting phenomena in metal-ceramic joining. We postulate the existence of a “solvent effect” due to the interaction between the reactive element addition and the brazing alloy. This effect plays a significant role in reactive wetting. By taking this effect into account, more realistic reactivities of different reactive element additions into a given brazing base alloy are predicted. Irreversible thermodynamics are also used to characterize the driving forces for reactive metal-ceramic joining.
Similar content being viewed by others
Abbreviations
- α:
-
equilibrium progressive factor
- γ:
-
activity coefficient
- Г:
-
surface excess
- ν, ξ:
-
chemical stoichiometries of compound
- Ξ:
-
=[ΔG o(ReXν - ΔH M(Re)/gn]
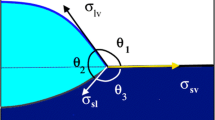
- σ:
-
surface tension
- μ:
-
chemical potential
- Π:
-
surface area
- ρ:
-
density
- Θ:
-
contact angle
- ω:
-
= (L 1w -L wgl Г i )R
- a :
-
activity
- G :
-
Gibbs energy
- R:
-
gas constant
- T :
-
temperature
- H M :
-
partial mole enthalphy of mixing
- x :
-
mole fraction
- S, s :
-
entropy, entropy per unit mass
- U, u :
-
internal energy, internal energy per unit mass
- p :
-
pressure
- V :
-
volume
- N, n :
-
moles of component, moles of component per unit mass
- v :
-
velocity
- t :
-
time
- J :
-
flux
- K :
-
rate of chemical reaction
- F :
-
body force
- g :
-
acceleration due to gravity
- A :
-
chemical affinity
- e :
-
surface area per unit mass
- L :
-
phenomenological coefficient
References
I.A. Aksay, C.E. Hoge, and J.A. Pask:J. Phys. Chem., 1974, vol. 78, p. 1178.
J.E. McDonald and J.G. Eberhart:TMS-AIME, 1965, vol. 233, p. 512.
D. Chatain, I. Rivollet, and N. Eustathopoulos:J. Chim. Phys., 1986, vol. 83, p. 561.
F.G. Yost and A.D. Romig, Jr.:Mater. Res. Soc. Symp. Proc, 1988, vol. 108, p. 385.
P.R. Chidambaram, G.R. Edwards, and D.L. Olson:Metall. Trans. B, 1992, vol. 23B, pp. 215–22.
E.T. Turkdogan:Physical Chemistry of High Temperature Technology, Academic Press, New York, NY, 1980, p. 93.
R.R. Kapoor and T.W. Eagar:Metall. Trans. B, 1989, vol. 20B, pp. 919–24.
R.R. Kapoor and T.W. Eagar:Ceramic Engineering and Science Proceedings, American Ceramic Society, Westerville, OH, 1989, p. 1613.
M. Naka and I. Okamoto:Trans. JWRI, 1985, vol. 14, pp. 29–34.
A.R. Miedema, F.R. de Boer, and R. Boom:CALPHAD, 1977, vol. 1, p. 341.
C. Wagner:Thermodynamics of Alloys, Addison-Wesley Press Inc., Cambridge, MA, 1952, p. 25.
L.B. Pankrantz, J.M. Stuve, and N.A. Gokcen:Thermodynamic Data for Mineral Technology, U.S. Bureau of Mines Bull. 677, U.S. Government Printing Office, Washington, DC, 1984.
J.J. Pak, M.L. Santella, and R.J. Fruehan:Metall. Trans. B, 1990, vol. 21B, pp. 349–55.
P. Glasdorff and I. Prigogine:Physica, 1964, vol. 30, p. 351.
R.B. Bird, W.E. Steward, and E.N. Lightfoot:Transport Phenomena, John Wiley & Sons, Inc., New York, NY, 1960.
S.R. de Groot and P. Mazur:Non-Equilibrium Thermodynamics, North Holland Publishing Co., Amsterdam, 1962.
T. Young:Phil. Trans. R. Soc. London, 1805, vol. 95, p. 65.
F. Brochard-Wyart and P.G. de Gennes:Adv. Colloid Interface Sci., 1992, vol. 39, p. 1.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, G., Lannutti, J.J. Chemical thermodynamics as a predictive tool in the reactive metal brazing of ceramics. Metall Mater Trans A 26, 1499–1505 (1995). https://doi.org/10.1007/BF02647601
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02647601