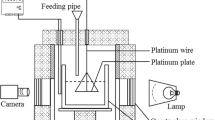
Abstract
Alumina powder dissolves rapidly in molten cryolite,i.e. in 4 to 8 seconds, provided that the particles are effectively dispersed in the melt. At alumina contents well below saturation the time of dissolution is independent of the alumina concentration, and it is not appreciably affected by additions of NaF, A1F3, or CaF2. Coarse grains dissolve more slowly than fine grains and α-Al2O3 (floury) dissolves more slowly than γ-Al2O3 (sandy). The rate-controlling step appears to be a slow chemical reaction. Bottom sludge in industrial cells contains about 40 pct A12O3. This material has a higher α/γ-ratio than the alumina being fed to the cells, while the grain size is smaller due to disintegration of aggregates. The time of dissolution is identical to that of any other alumina with the same α/γ-ratio.
Similar content being viewed by others
References
N. Parravano and O. D’Agostino:Atti Reale Accad. Lincei, 1932, pp. 186-90.
G. A Abramov and N. A. Kaluzhkii:Tsvet. Met, 1952, vol. 25, no. 4, pp. 31- 40.
J. L. Henry and W. M. Lafky:Ind. Eng. Chem., 1956, vol. 48, pp. 126–28.
K. G. Marin and A. I. Belyaev:Sb. Nauch. Tr. Moscow Inst. Tsvet. Met. Zolota, 1957, no. 27, pp. 178-92.
E. A. Zhemchuzhina and A. I. Belyaev:Nauch. Dokl. Vyssh. Shk. Met, 1959, vol. 2, no. 2, pp. 56–60.
A. I. Belyaev:The Electrolyte of Aluminum Cells, pp. 129–37, Metallurgizdat, Moscow, 1969.
L. A. Firsanova and A. I. Belyaev:Izv. Vyssh. Ucheb. Zaved., Tsvet. Met, 1962, vol. 5, no. 1, pp. 77–81.
L. N. Antipin, S. F. Vazhenin, V. T. Verkhovets, and V. V. Nerubashenko:Tr. Vses. Soveshch. po Fiz. Khim., Rasplav. Solei, 2nd, Kiev, 1963; pp. 95-98,Metallurgiya, Moscow, 1965.
H. Latreille:Electrochim. Metal, 1968, vol. 3, pp. 359–68.
I. S. Kachanovskaya, N. S. Siraev, and N. V. Pototskaya:Tsvet. Met, 1969, vol. 42, no. 9, pp. 64–66.
G. Winkhaus:TMS Paper Selection Program, no. A70-25,1970.
N. M. Drobot and T. I. Olgina:Tsvet. Met, 1971, vol. 44, no. l,pp. 38–40.
L. V. Biriukova:Tsvet. Met, 1956, vol. 29, no. 1, pp. 57–61.
F. Mosoczi:Kohasz. Lapok, 1956, vol. 11, pp. 465–71.
E. Becker, G. Paczolay, and O. Klug:Femip. Kut Intez. Kozlem., 1959, vol. 3, pp. 69–73.
S. Bretsznajder, J. Pysiak, and P. Krzyzanowski:Przetny. Chem., 1964, vol. 43 no. 6, pp. 299–302.
E. Balazs and J. Adam:Freiberg. Forschungsh., 1965, vol. B103, pp. 91–101.
Int. Stand. Org., ISO/TC47/GT48, USA 51, no. 476.
Int. Stand. Org, ISO/TC47/WG8, Germany 34, May 1969, no. 462.
K. Grjotheim, C. Krohn, and J. Thonstad: inInt. Leichtmetalltag., 5th, Leoben pp. 343–46, Aluminum-Verlag, Düsseldorf, 1968.
J. Thonstad:Electrochim. Acta, 1969, vol. 14, pp. 127–34.
P. M. Shurygin, V. N. Boronenkov, and V. I. Kryuk:Izv. Vyssh. Uchebn. Zaved., Tsvet. Met., 1962, vol. 5, no. 3, pp. 59–66.
P. M. Shurygin, L. M. Barmin, and V. N. Boronenkov:Izv. Vyssh. Uchebn. Zaved., Tsvet. Met, 1962, vol. 5, no. 4, pp. 106–12.
P. M. Shurygin, V. N. Boronenkov, and V. 1. Kryuk:Sb. Nauch. Tr. Uralsk. Politechn. Inst, 1963, vol. 126, pp. 80–85.
T. Yokokawa and O. J. Kleppa:J Phys. Chem., 1964, vol. 68, pp. 3246–48.
V. J. Hill: inExtractive Metallurgy of Aluminum, vol. 1, pp. 219–28, G. Gerard ed., Interscience Publ., New York, 1963.
E. W. Dewing:Trans. TMS-AIME, 1969, vol 245, pp. 2181–84.
N. W. F. Phillips, R. H. Singleton, and E. A. Hollingshead:J. Electrochem. Soc, 1955, vol. 102, pp. 648–49.
A. Fenerty and E. A. Hollingshead:J. Electrochem. Soc, 1960, vol. 107, pp. 993–97.
M. F. Landi, R. Bacchiega, and A. Battaglia:Metallurgia Ital., 1968, vol. 60, pp. 939–44.
J. W. Fisher:Angew. Chem., 1948, vol. 20, pp. 17–23.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thonstad, J., Nordmo, F. & Paulsen, J.B. Dissolution of alumina in molten cryolite. Metall Trans 3, 407–412 (1972). https://doi.org/10.1007/BF02642044
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02642044