Summary
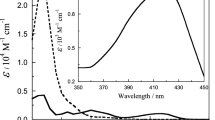
The preparation of a solution of sodium gossypolate in aqueous acetone has been described. Indications of a 1∶1 mole ratio combination of the gossypolate and ferrous ions were established through conductometric and potentiometric studies in the presence of either chloride or perchlorate anion. Application of the method of continuous variations to differences in the absorption spectra of the two species confirmed the 1∶1 mole ratio of ferrous ion to gossypol. This finding is supported by analytical data. Values of pK for the dissociation constant of ferrous gossypolate, calculated from spectrophotometric measurement, averaged 7.3.
Since the peri-hydroxyls are the most plausible location of the sodium ions in sodium gossypolate, its reaction with ferrous ion results in displacement of the two sodium ions by the ferrous ion.
Similar content being viewed by others
References
Adams, R., Morris, R. C., Geissman, T. A., Butterbaugh, D. J., and Kirkpatrick, E. C., J. Am. Chem. Soc.,60, 2193–2204 (1938).
Carruth, F. E., J. Am. Chem. Soc.,40, 647–663 (1918).
Gallup, W. D., J. Biol. Chem.,77, 437–449 (1928).
Job, P., Ann. chim., [10]9, 113–203 (1928).
Marchlewski, L., J. prakt. Chem.,60, 84–90 (1899).
Turner, S. E., and Anderson, R. C., J. Am. Chem. Soc.,71, 912–914 (1949).
Withers, W. A., and Brewster, J. F., J. Biol. Chem.15, 161–166 (1913).
Withers, W. A., and Carruth, F. E., J. Agr. Research,5, 261–288 (1915).
Withers, W. A., and Carruth, F. E., J. Biol. Chem.,32, 245–257 (1917).
Author information
Authors and Affiliations
Additional information
Findings reported in this paper were included in a thesis submitted to Tulane University by Robert J. Demint in partial fulfillment of the requirements for the M.S. degree.
One of the laboratories of the Southern Utilization Research Branch, Agricultural Research Service, U. S. Department of Agriculture.
About this article
Cite this article
Jonassen, H.B., Demint, R.J. Interaction of gossypol with the ferrous ion. J Am Oil Chem Soc 32, 424–426 (1955). https://doi.org/10.1007/BF02639702
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02639702