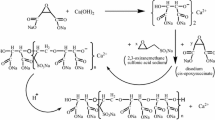
Conclusion
Detergents prepared from Fischer-Tropsch olefins are thus measurably better than the reference standards prepared from propylene tetramer or lauryl alcohol. The combination of straight and branched chains with both terminal and internal double bonds produces the high level of surface activity found with these synthetic olefins.
Although the optimum molecular weights for the experimental sulfonates are about the same as for sulfonates from polymer olefins, the surface activity is somewhat better. In physical properties these sulfonates are superior to polymer-olefin sulfonates in resistance to caking and in rate of solution. The optimum experimental sulfates, of 14- through 16-carbon atoms, are superior to lauryl sulfate. The branched structure of these alkyl sulfates probably gives rise to their soft, waxy, and hygroscopic character. Total mixtures of these experimental products are better than the respective reference standards.
Both types of detergents prepared from Fischer-Tropsch olefins are about equivalent. The alkylbenzene sulfonates appear to be superior to similar products now in every-day use. Although Fischer-Tropsch olefins are a promising source of raw material for detergents, there is no assurance that they will be commercially available in the near future.
Similar content being viewed by others
References
Adams, C. E., Proell, W. A., and Ballweber, E. G. (Standard Oil Co. [Ind.]), U. S. 2,585,983 (1954).
American Association of Textile Chemists and Colorists, “Technical Manual and Year Book,” vol. 27, p. 142–144, New York, Howes Publishing Company Inc., 1951.
Am. Soc. Testing Materials, Standards on Soaps and Other Detergents, D-12, D 1173–51T (1953).
Antsus, L. I., and Petrov, A. D., Zhur. Obshchei. Khim,3, 253 (1933).
Bertsch, H. (American Hyalsol Corporation), U. S. 1,968,793-7 (1934).
Bramston-Cook, H. E., and Elwell, W. E., Ind. Eng. Chem.,46 1922–1924 (1954).
Crowe, J. B., Am. Soc. Testing Materials, Proc.,43, 378–384 (1943).
Field, E. (Standard Oil Co. [Ind.]), U. S. 2,683,177 (1954).
Holm, M. M.et al. “The Oxo Process” (FIAT Final Rept. 1000) Washington, D. C., Off. of Tech. Services, U. S. Dept. of Commerce (1947).
Kircher, J. E., Miller, E. L., and Geiser, P. E., Ind. Eng. Chem.,46, 1925–1930 (1954).
Lien, A. P., Hill, P., and Deters, J. F. (Standard Oil Co. [Ind.]), U. S. 2,564,072 (1951).
Roelen, Otto (Alien Prop. Custodian), U. S. 2,327,066 (1943).
Shedlovsky, L., Ann. N. Y. Acad. Sci.,46, Art. 6, 427 (1946).
Smith, D. F., Hawk, C. O., and Golden, P. L., J. Am. Chem. Soc.,52, 3221–3232 (1930).
U. S. Bur. Mines Inform. Circ. 7370, 87–92 (1946).
Vaughan, T. H., and Smith, C. E., J. Am. Oil Chem. Soc.,25, 44–51 (1948).
Weitkamp, A. W., and Frye, C. G., Ind. Eng. Chem.,45, 363–367 (1953).
Weitkamp, A. W., Seelig, H. S., Bowman, N. J., and Cady, W. E., Ind. Eng. Chem.,45, 343–349 (1953).
Author information
Authors and Affiliations
About this article
Cite this article
Ravenscroft, P.H., Turney, M.E. Detergents from synthetic olefins. J Am Oil Chem Soc 32, 418–420 (1955). https://doi.org/10.1007/BF02639699
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02639699