Summary
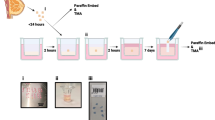
Normal human endometrial cells were grown on an extracellular matrix containing type IV collagen, laminin, heparan sulfate proteoglycan, and entactin (Matrigel). On the extracellular matrix, dispersed endometrial cells remained rounded, and aggregated to form mounds of cells, which continued to grow in this arrangement. At 10 d, light microscopy demonstrated that these mounds were comprised of an eosinophilic substance, containing individual fusiform stromal cells. About 50% of the mounds were covered with a single layer of polarized cuboidal to columnar cells with basal nuclei, whereas 60% contained columnar cells forming glandular structures with open lumina. These polarized cuboidal and columnar cells were epithelial, based on their positive staining for cytokeratins and the possession of microvilli, tonofilaments, abundant glycogen, ribosomes, and primitive junctional complexes. Kreyberg's stain showed the presence of mucin within the lumina of the glands, indicating that they were functional. Thus, human endometrial cells grown on an extracellular matrix form a simple cuboidal to columnar epithelium, a stromal component, and glandular structures, thereby mimicking the in vivo morphology of the endometrium.
Similar content being viewed by others
References
Burwen, S.; Pitelka, D. R. Secretory function of lactating mouse mammary epithelial cells cultured on collagen gels. Exp. Cell Res. 126:249–262; 1980.
Carson, D. D.; Tang, J.-P.; Julian, J., et al. Vectorial secretion of proteoglycans by polarized rat uterine epithelial cells. J. Cell. Biol. 107:2425–2435; 1988.
Centola, G. M.; Cisar, M.; Knab, D. R. Establishment and morphologic characterization of normal human endometrium in vitro. In Vitro 20:451–461; 1984.
Churukian, C. J.; Schenk, E. A. A modification of Kreyberg's method for demonstrating keratin and mucin. J. Histotechnol. 7:146; 1984.
di Sant'Angese, P.A.; De Mesy-Jensen, K. L. Diagnostic electron microscopy on reembedded (“popped off”) areas of large spurr epoxy sections. Ultrastruct. Pathol. 6:247–253; 1984.
Eckert, R. L., Katzenellenbogen, B. S. Human endometrial cells in primary tissue culture: modulation of the progesterone receptor level by natural and synthetic estrogensin vitro. J. Clin. Endocrin. Metab. 52:699–708; 1981.
Emerman, J. T.; Enami, J.; Pitelka, D. R., et al. Hormonal effects on intracellular and secreted casein in cultures of mouse mammary epithelial cells on floating collagen membranes. Proc. Natl. Acad. Sci. USA 74:4466–4470; 1977.
Emerman, J. T.; Pitelka, D. R. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro 13:316–328; 1977.
Fortier, M. A.; Guilbault, L. A.; Grasso, F. Specific properties of epithelial and stromal cells from the endometrium of cows. J. Reprod. Fertil. 83:239–248; 1988.
Frauli, M.; Ludwig, H. Inhibition of fibroblast proliferation in a culture of human endometrial stromal cells using a medium containingd-Valine. Arch. Gynecol. Obstet. 241:87–96; 1987.
Gerschenson, L. E.; Berliner, J.; Yang, J.-J. Diethylstilbestrol and progesterone regulation of cultured rabbit endometrial cell growth. Cancer Res. 34:2873–2880; 1974.
Glasser, S. R. Laboratory models of implantation. In: Dixon, R. L., ed. Reproductive toxicology. New York: Raven Press; 1985:219–237.
Glasser, S. R.; Julian, J.; Decker, G. L., et al. Development of morphological and functional polarity in primary cultures of immature rat uterine epithelial cells. J. Cell. Biol. 107:2409–2423; 1988.
Glasser, S. R.; Julian, J.; Mani, S. K.; et al. Blastocyst-endometrial relationships: reciprocol interactions between uterine epithelial and stromal cells and blastocysts. Trop. Res. 5: in press.
Goldin, G. V. Towards a mechanism for morphogenesis in epitheliomesenchymal organs. Rev. Biol. 55:251–265; 1980.
Gospodarowicz, D.; Greenburg, G.; Birdwell, C. R. Determination of cellular shape by the extracellular matrix and its correlation with the control of cellular growth. Cancer Res. 38:4155–4171; 1978.
Hadley, M. A.; Byers, S. W.; Suarez-Quian, C. A., et al. Extracellular matrix regulates sertoli cell differentiation, testicular cord formation, and germ cell development in vitro. J. Cell Biol. 101:1511–1522; 1985.
Iguchi, T.; Uchima, F.-D.; Ostrander, P. L., et al. Growth of normal mouse vaginal epithelial cells in and on collagen gels. Proc. Natl. Acad. Sci. USA 80:3743–3747; 1983.
Kassis, J. A.; Sakai, D.; Walent, J. H., et al. Primary cultures of estrogen-responsive cells from rat uteri: induction of progesterone receptors and a secreted protein. Endocrinology 114:1558–1566; 1984.
Kleinman, D.; Sharon, Y.; Sarov, I., et al. Human endometrium in cell culture: a new method for culturing human endometrium as separate epithelial and stromal components. Arch. Gynecol. 234:103–112; 1983.
Kreyberg, L. Mainm histological types of primary epithelial lung tumors. Br. J. Cancer 25:206–210; 1961.
Lunginbuhl, W. H. Electron microscopic study of the effects of tissue culture on human endometrium. Am. J. Obstet. Gynecol. 102:192–201; 1968.
Maslar, I. A.; Ansbacher, R. Effect of short-duration progesterone treatment on decidual prolactin production by cultures of proliferative human endometrium. Fertil. Steril. 50:250–254; 1988.
Mulholland, J.; Winterhager, E.; Beier, H. M. Changes in proteins synthesized by rabbit endometrial epithelial cells following primary culture. Cell Tissue Res. 252:123–132; 1988.
Pavlik, E. J.; Katzenellenbogen, B. S. Human endometrial cells in primary tissue culture: Estrogen interactions and modulation of cell proliferation. J. Clin. Endocrin. Metab. 47:333–344; 1978.
Rinehart, C. A.; Lyn-Cook, B. D.; Kaufman, D. G. Gland formation from human endometrial epithelial cells in vitro. In Vitro Cell. Dev. Biol. 24:1037–1041; 1988.
Satyaswaroop, P. G.; Bressler, R. S.; de la Pena, M. M., et al. Isolation and culture of human endometrial glands. J. Clin. Endocrinol. Metab. 48:639–641; 1979.
Sengupta, J.; Given, R. L.; Carey, J. B.; et al. Primary culture of mouse endometrium on floating collagen gels: a potential in vitro model for implantation. Ann. NY Acad. Sci. 476:75–94; 1986.
Shannon, J. M.; Pitelka, D. R. The influence of cell shape on the induction of functional differentiation in mouse mammary cells in vitro. In Vitro 17:1016–1028; 1981.
Trent, J. M.; Davis, J. R.; Payne, C. M. The establishment and morphological characterization of finite cell lines from normal human endometrium. Am. J. Obstet. Gynecol. 136:352–362; 1980.
Varma, V. A.; Melin, S. A.; Adamec, T. A., et al. Monolayer culture of human endometrium: methods of culture and identification of cell types. In Vitro 18:911–918; 1982.
Watt, F. M.; Jordan, P. W.; O'Neill, C. H. Cell shape controls terminal differentiation of human epidermal keratinocytes. Proc. Natl. Acad. Sci. USA 85:5576–5580; 1988.
Wèidmann, J.; Freund, M.; McGeever-Rubin, B. Cytologic observations on trypsinized cells obtained from human endometrial epithelium in tissue culture. J. Histotechnol. 10:233–235; 1987.
White, T. E. K.; Miller, R. K.; Grudzinskas, G., et al. Human endometrial cells grown on extracellular matrix (ECM) form an in vivo type morphology and produce the endometrial protein PP-14 (α2-PEG). Placenta 10:520; 1989.
Yang, J.; Flynn, D.; Larson, L.; et al. Growth in primary culture of mouse submandibular epithelial cells imbedded in collagen gels. In Vitro 18:435–442; 1982.
Author information
Authors and Affiliations
Additional information
This work was supported by grants ES02774, ES01247 and ES07026 (an NIEHS Toxicology Training Grant awarded to T.W for doctoral studies), from the National Institutes of Health, Bethesda, MD. Portions of this work were presented at the 1988 meetings of the Society for the Study of Reproduction in Seattle, Washington, and the Eleventh Rochester Trophoblast Conference.
Rights and permissions
About this article
Cite this article
White, T.E.K., di Sant'agnese, P.A. & Miller, R.K. Human endometrial cells grown on an extracellular matrix form simple columnar epithelia and glands. In Vitro Cell Dev Biol 26, 636–642 (1990). https://doi.org/10.1007/BF02624214
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02624214