Summary
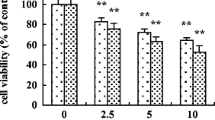
The effect of Zn-induced metallothionein (MT) on the toxicity, uptake, and subcellular distribution of cadmium (Cd) was examined in rat primary hepatocyte cultures and compared to results obtained earlier in this laboratory from intact animals. Hepatocytes were isolated and grown in monolayer culture for 22 h and subsequently treated with ZnCl2 (100 μM) for 24 h, which increased MT concentration about 15-fold. After Zn pretreatment, hepatocytes were exposed to Cd for 24 h. Cytotoxicity was assessed by enzyme leakage, intracellular potassium loss, and cellular glutathione content. The toxicity of Cd was much less in Zn-pretreated cells than in control cells, similar to that previously demonstrated in the intact animal. Zn pretreatment had no appreciable effect on the hepatocellular uptake of109Cd, but markedly altered its subcellular distribution, with more Cd accumulating in the cytosol and less in the nuclear, mitochondrial, and microsomal fractions. In the cytosol of Zn-pretreated cells, Cd was associated mainly with MT; in contrast, cytosolic Cd in control cells was mainly associated with non-MT macromolecules. Zn-induced changes in the subcellular distribution of Cd in vitro are identical to those observed in vivo in Zn-pretreated rats challenged with Cd. In summary, Zn pretreatment of rat primary hepatocyte cultures protects cells against Cd toxicity. Protection seems to be due to MT-promotes sequestration of Cd and reduction of the amount of Cd associated with critical organelles and proteins. These observations are similar to those noted in the whole animal. These results indicate that cultured hepatocytes are an ideal model for examining MT-induced tolerance to Cd hepatotoxicity.
Similar content being viewed by others
References
Berry, M. N.; Friend, D. S. High-yield preparation of isolated rat liver parenchymal cells. J. Cell Biol. 43:506–520; 1969.
Bissell, D. M.; Guzelian, P. S. Phenotypic stability of adult rat hepatocytes in primary monolayer culture. Ann. NY Acad. Sci. 349:85–98; 1980.
Bracken, W. M.; Klaassen, C. D. Induction of metallothionein by steroids in rat primary hepatocyte cultures: evidence for direct induction. Toxicol. Appl. Pharmacol. 22:163–174; 1987.
Bracken, W. M.; Klaassen, C. D. Induction of hepatic metallothionein by alcohols: evidence for an indirect mechanism. Toxicol. Appl. Pharmacol. 87:257–263; 1987.
Bracken, W. M.; Klaassen, C. D. Induction of metallothionein in rat primary hepatocyte cultures: evidence for direct and indirect induction. J. Toxicol. Environ. Health. 22:163–174; 1987.
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254; 1976.
Cantilena, L. R.; Klaassen, C. D. Isolated rat hepatocytes as a model for screening chelators for use in cadmium intoxication. Toxicol. Appl. Pharmacol. 67:361–367; 1983.
Din, W. S.; Frazier, J. M. Protective effect of metallothionein on cadmium toxicity in isolated rat hepatocytes. Biochem. J. 230:395–402; 1985.
Dudley, R. E.; Svoboda, D. J.; Klaassen, C. D. Acute exposure of cadmium causes severe liver injury in rats. Toxicol. Appl. Pharmacol. 65:302–313; 1982.
Eaton, D. L.; Toal, B. F. Evaluation of the Cd/hemoglobin affinity assay for the rapid determination of metallothionein in biological tissues. Toxicol. Appl. Pharmacol. 66:134–142; 1982.
Failla, M. L.; Cousins, R. J.; Mascenik, M. J. Cadmium accumulation and metabolism by rat liver parenchymal cells in primary monolayer culture. Biochim. Biophys. Acta. 583:63–72; 1979.
Goering, P. L.; Klaassen, C. D. Zinc-induced tolerance to cadmium hepatotoxicity. Toxicol. Appl. Pharmacol. 74:299–307; 1984.
Goering, P. L.; Klaassen, C. D. Tolerance to cadmium-induced hepatotoxicity depends on presynthesized metallothionein in liver. J. Toxicol. Environ. Health. 14:803–812; 1984.
Henry, R. T.; chiamori, N.; Golub, O. J., et al. Spec-trophotometric methods for the determination of glutamic-oxalacetic transaminase, glutamic-pyruvic transaminase and lactic dehydrogenase. Am. J. Clin. Pathol. 34:381–398; 1960.
Holme, J. A. Xenobiotic metabolism and toxicity in primary monolayer cultures of hepatocytes. NIDH Annals 8:49–63; 1985.
Ichihara, A.; Nakamura, T.; Tanaka, K., et al. Biochemical functions of adult rat hepatocytes in primary culture. Ann. NY Acad. Sci. 349:77–84; 1980.
Klaassen, C. D.; Bracken, W. M.; Dudley, R. E., et al. Role of sulfhydryls in the hepatotoxicity of organic and metallic compounds. Fund. Appl. Toxicol. 5:806–815; 1985.
Kotsonis, F. N.; Klaassen, C. D. Toxicity and distribution of cadmium administered to rats at sublethal doses. Toxicol. Appl. Pharmacol. 41:667–680; 1977.
Kudo, N.; Yamashina, S.; Waku, K. Protection against cadmium toxicity by zinc: decrease in the Cd-high molecular weight protein fraction in rat liver and kidney on Zn pretreatment. Toxicology 40:267–277; 1987.
Lehman, L. D.; Klaassen, C. D.. Dosage-dependent disposition of cadmium administrated orally to rats. Toxicol. Appl. Pharmacol. 84:159–167; 1986.
Müller, L. Consequence of cadmium toxicity in rat hepatocytes: mitochondrial dysfunction and lipid peroxidation. Toxicology 40:285–295; 1986.
Müller, L; Wilhelm, M. Effects of cadmium in rat hepatocytes: interaction with aluminum. Toxicology 44:193–291; 1987.
Nakamura, T.; Ichihara, A. Control of growth and expression of differentiated functions of mature hepatocytes in primary culture. Cell Struct. Funct. 10:1–16; 1985.
Santone, K. S.; Acosta, D. Cadmium toxicity in primary cultures of rat hepatocytes. J. Toxicol. Environ. Health. 10:169–177; 1985.
Stacey, N. H.; Klaassen, C. D. Cadmium uptake by isolated rat hepatocytes. Toxicol. Appl. Pharmacol. 55:448–455; 1980.
Stacey, N. H.; Cantilena, L. R.; Klaassen, C. D. Cadmiuminduced and lipid peroxidation in isolated rat hepatocytes. Toxicol. Appl. Pharmacol. 53:470–480; 1980.
Stacey, N. H. Protective effects of dithiothreitol on cadmium-induced injury in isolated rat hepatocytes. Toxicol. Appl. Pharmacol. 82:226–232; 1986.
Stacey, N. H.; Klaassen, C. D. The amelioration of cadmium-induced injury in isolated hepatocytes by reduced glutathione. Toxicology 42:85–93; 1986.
Steel, C. D.; Torrie, J. H. Principles and procedures of statistics. New York: McGraw-Hill Book Co.; 1960; 99–160.
Stein, A. F.; Bracken, W. M.; Klaassen, C. D. Utilization of methionine as a sulfhydryl source for metallothionein synthesis in rat primary hepatocyte cultures. Toxicol. Appl. Pharmacol. 87:267–283; 1987.
Tietze, F. Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione. Anal. Biochem. 27:502–522; 1960.
Waalkes, M.; Klaassen, C. D. Concentration of metallothionein in major organs of rats after administration of various metals. Fund. Appl. Toxicol. 5:473–477; 1985.
Webb, M. Role of metallothionein in cadmium metabolism. In: Foulkes, E. C., ed. Cadmium handbook of experimental pharmacology, vol. 80. Berlin, Heidelberg; Springer-Verlag Press; 1986:281–328.
Wong, K. L.; Klaassen, C. D. Relationship between tissue levels of glutathione and metallothionein in rats. Toxicology 19:39–47; 1981.
Author information
Authors and Affiliations
Additional information
This work was supported by grant ES-01142, and WCK was supported by training grant ES-07079, both from the Public Health Service, Department of Health and Human Services.
Rights and permissions
About this article
Cite this article
Liu, J., Kershaw, W.C. & Klaassen, C.D. Rat primary hepatocyte cultures are a good model for examining metallothionein-induced tolerance to cadmium toxicity. In Vitro Cell Dev Biol 26, 75–79 (1990). https://doi.org/10.1007/BF02624158
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02624158