Summary
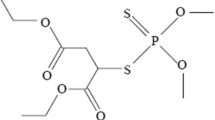
Neurons from brains of chick embryos and pond snails (Lymnaea stagnalis) were cultured for 3 to 4 d in the presence of no toxins, inorganic lead (PbCl2), or organic lead (trielthyl lead chloride). In chick neurons, inorganic lead reduced the percentage of cells that grew neurites (IC50=270μM total lead, approximately 70 nM free Pb2+) but did not reduce the number of neurites per cell or the mean neurite length. Triethyl lead reduced the percentage of cells that grew neuites (IC50=0.24 μM) and the mean neurite length (extrapolated IC50=3.6 μM) but did not reduce the number of neurites per cell. InLymnaea neurons, inorganic lead reduced the percentage of cells that grew neurites (IC50=13 μM total lead; approximately 10 nM free Pb2+). Triethyl lead reduced the percentage of cells that grew neurites (IC50=0.4 μM) and exerted significant toxicity at 0.2 μM. The two forms of lead affected neurite growth in qualitatively different ways, which suggests that their mechansms of action are different.
Similar content being viewed by others
References
Aletta, J. M.; Greene, L. A. Growth cone configuration and advance: a time-lapse study using video-enhanced differential interference contrast microscopy. J. Neurosci. 8:1425–1435; 1988.
Alfano, D. P.; Petit, T. L. Neonatal lead exposure alters the dendritic development of hippocampal dentate granule cells. Exp. Neurol. 75:275–288; 1982.
Alfano, D. P.; LeBoutillier, J. C.; Petit, T. L. Hippocampal mossy fiber pathway development in normal and postnatally lead-exposed rats. Exp. Neurol. 75:308–319; 1982.
Anglister, L.; Farber, I. C.; Shahar, A., et al. Localization of voltage-sensitive calcium channels along developing neurites: their possible role in regulating neurite elongation. Dev. Biol. 94:351–365; 1982.
Atchison, W. D.; Narahashi, T. Mechanisms of action of lead on neuromuscular junctions. Neurotoxicology 5:267–282; 1984.
Audesirk, G. Effects ofin vitro andin vivo, lead exposure on voltage-dependent calcium channels in central neurons ofLymnaea stagnalis. Neurotoxicology 8:579–592; 1987.
Audesirk, G.; Nelson, G.; Shugarts, D., et al. Inhibition of neurite growth by organic and inorganic lead. Soc. Neurosci. Abstr. 14:1265; 1988.
Buesselberg, D.; Evans, M. L.; Carpenter, D. O. The effect of lead on the voltage activated calcium channel ofAplysia neurons. Soc. Neurosci. Abstr. 14:1081; 1988.
Coates, P. W. Quantitation and morphological characterization of rapid axon and dendritic growth from single cerebral hemispheric neurons in hydrated collagen lattice culture. Dev. Brain Res. 25:11–20; 1986.
Cookman, G. R.; Hemmens, S. E.; Keane, G. J., et al. Chronic low level lead exposure precociously induces rat glial development in vitro and in vivo. Neurosci. Lett. 86:33–37; 1988.
Cooper, G. P.; Steinberg, D. Effects of cadmium and lead on adrenergic neuromuscular transmission in the rabbit. Am. J. Physiol. 232:C128-C131; 1977.
Cremer, J. E. Possible mechanisms for the selective neurotoxicity. In: Grandjean, P.; Grandjean, E. C. eds. Biological effects of organolead compounds. Boca Raton, FL: CRC Press: 1984:207–218.
Davis, J. M.; Svendsgaard, D. J. Dead and child development. Nature 329:297–300; 1987.
Dietrich, K. N. Krafft, K. M.; Bornschein, R. L., et al. Low-level fetal lead exposure effect on neurobehavioral developement in early infancy. Pediatrics 80:721–730; 1987.
Faulstich, H.; Stournaras, C.; Doenges, K H., et al. The molecular mechanism of interaction of Et3Pb+ with tubulin. FEBS Lett. 174:128–131; 1984.
Ferris, N. J.; Cragg, B. G. Orgaic lead and histological parameters of brain development. Acta Neuropathol. 63:306–312; 1984.
Gotti, C.; Cabrini, D.; Sher, E., et al. Effects of long-termin vitro exposure to aluminum, cadmium or lead on differentiation and cholinergic receptor expression in a human neuroblastoma cell line. Cell Biol. Toxicol. 3:431–440; 1987.
Grundt, I. K.; Ammitzboll, T.; Clausen, J. Triethyl lead treatment of cultured brain cells. Neurochem. Res. 6:193–201; 1981.
Hansen, E. H.; Ruzicka, J. Selectrode—the universal solid-state electrode. Part VIII. The solid-state lead (II) selectrode in lead (II) buffers and potentiometric titrations. Anal. Chem. Acta 72:365–373; 1974.
Hsu, L.; Jeng, A. Y.; Chen, K. Y. Induction of neurite outgrowth from chick embronic anglia explants by activators of protein kinase C. Neurosci. Lett. 99:257–262; 1989.
Kater, S. B.; Mattson, M. P.; Cohan, C., et al. Calcium regulation of the neuronal growth cone. Trends Neurosci. 11:315–321; 1988.
Kauppinen, R. A.; Komulainen, H.; Taipale, H. T. Chloridedependent uncoupling of oxidative phosphorylation by triethyllead and triethyltin increases cytosolic free calcium in guinea pig cerebral cortical synaptosomes. J. Neurochem. 51:1617–1625; 1988.
Kiraly, E.; Jones, D. G. Dendritic spine changes in rat hippocampal pyramidal cells after postnatal lead treatment: a Golgi study. Exp. Neurol. 77:236–239; 1982.
Kivalo, P.; Virtanen, R.; Wickstrom, K. et al. An evaluation of some commercial lead(II)-selective electrodes. Anal. Chim. Acta 87:401–409; 1976.
Kober, T. E.; Cooper, G. P. Lead competitively inhibits calciumdependent synaptic transmission in the bullfrog sympathetic ganglion. Nature 262:704–705; 1976.
Kostial, K.; Vouk, V. B. Lead ions and synaptic transmission in the superior cervical ganglion of the cat. Br. J. Pharmacol. 12:212–219; 1957.
Lefauconnier, J. M.; Hauw, J. J.; Bernard, G. Regressive or lethal lead encephalopathy in the suckling rat: correlation of lead levels and morphological findings. J. Neuropath. Exp. Neurol. 42:177–190; 1983.
Letourneau, P. C.; Shattuck, T. A.; Ressler, A. H. “Pull” and “push” in neurite elongation: observations of the effects of different concentrations of cytochalasin B and taxol. Cell Motil. Cytoskeleton 8:193–209; 1987.
Lomme, M.; Ferguson, C.; Shugarts, D., et al. L-type calcium channels may regulate neurite growth in chick embryo brain neurons. Soc. Neurosci. Abstr. 14:1128; 1988.
Louis-Ferdinand, R. T.; Brown, D. R.; Fiddler, S. F., et al. Morphometric and enzymatic effects of neonatal lead exposure in the rat brain. Toxicol Appl. Pharmacol. 43:351–360; 1978.
Manalis, R. S.; Cooper, G. P. Presynaptic and postynaptic effects of lead at the frog neuromuscular junction. Nature 243:354–356; 1973.
Manalis, R. S.; Cooper, G. P.; Pomeroy, S. L. Effects of lead on neuromuscular transmission in the frog. Brain Res. 294:95–109; 1984.
Markovac, J.; Goldstein, G. W. Picomolar concentrations of lead stimulate brain protein kinase C. Nature 334:71–73; 1988.
Mattson, M. P.; Dou, P.; Kater, S. B. Outgrowth-regulating actions of glutamate in isolated hippocampal pyramidal neurons. J. Neurosci. 8:2087–2100; 1988.
Mattson, M. P.; Kater, S. B. Calcium regulation of neurite elongation and growth cone motility. J. Neurosci. 7:4034–4043; 1987.
Means, A. R.; Lagace, L.; Guerriero, V., Jr., et al. Calmodulin as a mediator of hormone action and cell regulation. J. Cell. Biochem. 20:317–330; 1982.
Nachshen, D. A. Selectivity of the Ca binding site in synaptosome Ca channels. Inhibition of Ca infllux by multivalent metal ions. J. Gen. Physiol. 83:941–967; 1984.
Needleman, H. L. Lead at low dose and the behavior of children. Acta Psychiatr. Scand. 67(Suppl)303:26–37; 1983.
Needleman, H. L. Low level exposure in the fetus and young child. Neurotoxicology 8:389–394; 1987.
Nicklowitz, W. J. Ultrastructural effects of acute treaethyl lead poisoning on nerve cells of the rabbit brain. Environ. Res. 8:17–36; 1974.
Petit, T. L.; LeBoutillier, J. C. Effects of lead exposure during development on neocortical dendritic and synaptic structure. Exp. Neurol. 64:482–492; 1979.
Pickett, J. B.; Bornstein, J. C. Some effects of lead at mammalian neuromuscular junction. Am. J. Physiol. 246:C271-C276; 1984.
Pocock, G.; Simons, T. J. B. Effects of lead ions on events associated with exocytosis in isolated bovine adrenal medullary cells. J. Neurochem. 48:376–382; 1987.
Pounds, J. G.; Wright, R.; Morrison, D.; et al. Effect of lead on calcium homeostasis in the isolated rat hepatocyte. Toxicol. Appl. Pharmacol. 63:389–401; 1982.
Press, M. F. Lead encephalopathy in nonata Long-Evans rats: morphological studies. J. Neuropath. Exp. Neurol. 34:169–193; 1977.
Roderer, G.; Doenges, K. H. Influence of trimethyl lead and inorganic lead on thein vitro assembly of micortubles from mammalian brain. Neurotoxicology 4:171–180; 1983.
Schanne, F. A. X.; Dowd, T. L.; Gupta, R. K., et al. Measurement of intracellular calcium ion concentrations in NG108-15 cells by fluorine-19 nuclear magnetic resonance spectroscopy: a model for the study of lead neurotoxicity. Soc. Neurosci. Abstr. 14:1264; 1988.
Shellenberger, M. K. Effects of early lead exposure on neurotransmitter systems in the brain: A reriew. Neurotoxicology 5:177–212; 1984.
Silbergeld, E. Behavioral teratology of lead. In: Yanai, J., ed., Neurobehavioral teratology. New York: Elsevier; 1984:433–445.
Simons, T. J. B. Influence of lead ions on cation permeability in human red cell ghosts. J. Membr. Biol. 84:61–71; 1985.
Simons, T. J. B.; Pocock, G. Lead enters bovine adrenal medullary cells through calcium channels. J. Neurochem. 48:383–389; 1987.
Spence, I.; Drew, C.; Johnston, G. A. R., et al. Acute effects of lead at central synapses in vitro. Brain Res. 333:103–109; 1985.
Suarez-Isla, B. A.; Pelto, D. J.; Thompson, J. M., et al. Blockers of calcium permeability inhibit neurite extension and formation of neuromuscular synapses in cell culture. Dev. Brain Res. 14: 263–270; 1984.
Suzuki, T.; Fujii, T.; Tanaka, R. Calcium/calmodulin-dependent inhibition of microtubule assembly by brain synaptic junction. Neurochem. Res. 11:543–555; 1986.
Walsh, T. J.; McLamb, R. L.; Bondy, S. C., et al. Triethyl and trimethyl lead: effects on behavior, CNS morphology and concentrations of lead in the blood and brain of rat. Neurotoxicology 7:21–34; 1986.
Windebank, A. J. Specific inhibition of myelination by leadin vitro; comparison with arsenic, thallium, and mercury. Exp. Neurol. 94:203–212; 1986.
Wong R. G.; Hadley, R. D.; Kater, S. B., et al. Neurite outgrowth in molluscan organ and cell cultures: the role of conditioning factor(s). J. Neurosci. 1: 1008–1021; 1981.
Yamamoto, H.; Fukunaga, K.; Tanaka, E., et al. Ca2+- and calmodulin-dependent phosphorylation of microtubule-associated protein 2 and τ factor, and inhibition of microtubule assembly. J. Neurochem. 41:1119–1125; 1983.
Zimmermann, H.-P.; Doenges, K. H.; Roderer, G. Interaction of triethyl lead chloride with microtubules in vitro and in mammalian cells. Exp. Cell Res. 156: 140–152; 1985.
Zimmermann, H.-P.; Plagens, U.; Traub, P. Influence of triethyl lead on neurofilamentsin vivo andin vitro. Neurotoxicology 8:569–578; 1987.
Author information
Authors and Affiliations
Additional information
These experiments were supported by grants from the Environmental Protection Agency, Washington, DC, and the National Institutes of Environmental Health Science, Research Triangle Park, NC.
Rights and permissions
About this article
Cite this article
Audesirk, G., Shugarts, D., Nelson, G. et al. Organic and inogranic lead inhibit neurite growth in vertebrate and invertebrate neurons in culture. In Vitro Cell Dev Biol 25, 1121–1128 (1989). https://doi.org/10.1007/BF02621263
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02621263