Summary
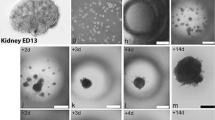
Interlobular and intralobular ducts isolated from the pancreas of the rat by digestion with collagenase and chymotrypsin were cultured in an agarose matrix containing CMRL-1066 supplemented with insulin, dexamethasone,l-glutamine, soybean trypsin inhibitor, antibiotics, and fetal bovine serum. The cut ends of most interlobular ducts sealed to create encolosed lumina. Some ducts retained their original cylindrical organization; others enlarged to varying degrees, resulting in structures that ranged from cylindrical to spherical in shape. The duct walls consisted of viable epithelium and connective tissue, although the amount of connective tissue declined with age. Both epithelial and connective tissue cells became flattened in the enlarged ducts. Intralobular and small interlobular ducts often remained associated with the larger interlobular ducts. These duct fragments have been cultured for as long as 6 weeks.
Similar content being viewed by others
References
Bolender, R. P. Stereological analysis of the guinea pig pancreas. I. Analytical model and quantitative description of non-stimulated pancreatic exocrine cells. J. Cell Biol. 61: 269–287; 1974.
Case, R. M. Secretory process in exocrine pancreas. Proc. Aust. Physiol. Pharmacol. Soc. 9: 29–42; 1978.
Singh, M.; Parks, N. M.; Webster, P. D., III. Anin vitro study of pancreatic ductal cells. Proc. Soc. Exp. Biol. Med. 157: 23–28; 1978.
Wizemann, V.; Christian, A-L.; Wiechmann, J.; Schulz, I. The distribution of membrane-bound enzymes in the acini and ducts of the cat pancreas. Pflugers Arch. 347: 39–47; 1974.
Jones, R. T.; Barrett, L. A.; van Haaften, C.; Harris, C. C.; Trump, B. F. Carcinogenesis in the pancreas. I. Long-term explant culture of human and bovine pancreatic ducts. J. Natl. Cancer Inst. 58: 557–565; 1977.
Lernmark, A.; Nathans, A.; Steiner, D. F. Preparation and characterization of plasma membrane-enriched fractions from rat pancreatic islets. J. Cell Biol. 71: 606–623; 1976.
Pictet, R.; Farsham, P. H. Long term islet culture (abstr.). Diabetes 24: 421; 1975.
Macpherson, I. Soft agar techniques. Kruse, P. F., Jr.; Patterson, M. K., Jr. eds. Tissue culture: methods and applications. New York: Academic Press; 1973: 276–280.
Barrett, L. A.; McDowell, E. M.; Frank, A. L.; Harris, C. H.; Trump, B. F. Long-term organ culture of human bronchial epithelium. Cancer Res. 36: 1003–1010; 1976.
Karnovsky, M. J. A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J. Cell Biol 27: 137A-138A; 1965.
Mollenhauer, H. H. Plastic embedding mixture for use in electron microscopy. Stain Technol. 39: 111–114; 1964.
Reynolds, E. S. The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 17: 208–212; 1963.
Githens, S.; Holmquist, D. R. G.; Whelan, J. F.; Ruby, J. R. Characterization of ducts isolated from the pancreas of the rat. J. Cell Biol. 85: 122–135; 1980.
Paul, J. Cell and tissue culture. London: Churchill Livingstone; 1975: 180–186.
Alwen, J.; Lawn, A. M. The reaggregation of adult rat liver cells maintained in vitro. Exp. Cell Res. 89: 197–205; 1974.
Leighton, J.; Mark, R.; Justh, G. Pattern of three-dimensional growth in vitro in collagen-coated cellulose sponge: carcinomas and embryonic tissues. Cancer Res. 28: 286–296; 1968.
Russo, J.; Bradley, R. H.; McGrath, C.; Russo, I. H. Scanning and transmission electron microscopic study of a human breast carcinoma line (MCF-7) cultured in collagen-coated cellulose sponge. Cancer Res. 37: 2004–2014; 1977.
Douglas, W. H. J.; Moorman, G. W.; Teel, R. W. The formation of histotypic structures from monodisperse fetal rat lung cells cultured on a three-dimensional substrate. In Vitro 12: 373–381; 1976.
Michalopoulos, G.; Pitot, H. C. Primary culture of parenchymal liver cells on collagen membranes. Morphological and biochemical observations. Exp. Cell Res. 94: 70–78; 1975.
Emerman, J. T.; Pitelka, D. R. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro 13: 316–328; 1977.
Hodges, G. M.; Melcher, A. H. Chemically-defined medium for growth and differentiation of mixed epithelial and connective tissues in organ culture. In Vitro 12: 450–459; 1976.
Schulz, I.; Yamagata, A.; Weske, M. Micropuncture studies on the pancreas of the rabbit. Pflügers Arch. 308: 277–290; 1969.
Fölsch, U. R.; Creutzfeldt, W. Pancreatic duct cells in rats: secretory studies in response to secretin, cholecytokinin-pancreozymin, and gastrin in vivo. Gastroenterology 73: 1053–1059; 1977.
Lightwood, R.; Reber, H. A. Micropuncture study of pancreatic secretion in the cat. Gastroenterology 72: 61–66; 1977.
Brecher, S. The occurrence and possible role of 80–100 Ä filaments in PtKI cells. Exp. Cell Res. 96: 303–310; 1975.
Githens, S.; Pictet, R.; Phelps, P.; Rutter, W. J. 5-Bromodeoxyridine may alter the differentiative program of the embryonic pancreas. J. Cell Biol. 71: 341–356; 1976.
Metz, J.; Merlo, M.; Billich, H.; Forssman, W. G. Exocrine pancreas under experimental conditions. IV. Alterations of intercellular junctions between acinar cells following pancreatic duct ligation. Cell Tissue Res. 186: 227–240; 1978.
Churg, A.; Richter, W. R. Early changes in the exocrine pancreas of the dog and rat after ligation of the pancreatic, duct. A light and electron microscopic study. Am. J. Pathol. 63: 521–546; 1971.
Cubilla, A. L.; Fitzgerald, P. J. Morphological patterns of primary nonendocrine human pancreas carcinoma. Cancer Res. 35: 2234–2248; 1975.
Pour, P.; Althoff, J.; Takahashi, M. Early lesions of pancreatic ductal carcinoma in the hamster model. Am. J. Pathol. 88: 291–308; 1977.
Levitt, M. H.; Harris, C. C.; Squire, R.; Springer, S.; Wenk, M.; Mollelo, C.; Thomas, D.; Kingsbury, E.; Newkirki, C. Experimental pancreatic carcinogensis. I. Morphogenesis of pancreatic adenocarcinoma in the Syrian golden hamster induced byN-nitroso-bis (2-hydroxypropyl) amine. Am. J. Pathol. 88: 5–28; 1977.
Author information
Authors and Affiliations
Additional information
This study was supported by National Cancer Institute Grant CA 19177 through the National Pancreatic Cancer Project and by Biomedical Research Support Grant RR 07196 from the Division of Research Resources, National Institutes of Health.
Rights and permissions
About this article
Cite this article
Githens, S., Holmquist, D.R.G., Whelan, J.F. et al. Ducts of the rat pancreas in agarose matrix culture. In Vitro 16, 797–808 (1980). https://doi.org/10.1007/BF02619315
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02619315