Abstract
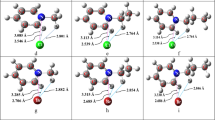
New types of ligands for spherical ions have been designed as part of the molecular design strategy for supramolecular compounds. In particular, symmetric conformers of macrocycles, built of −(CH2)n−O−(n=3,4,5) fragments, which envelope the spherical ion in a complementary way, have been constructed. Alos, new types of “bracelentands” with different, “hardening” fragments are suggested. Using the molecular mechanics methods, the selectivity of complex formation of the ligands with respect to alkaline metal ions has been investigated.
Similar content being viewed by others
References
V. E. Kuzmin and D. V. Pozigun,Zh. Fiz. Khim.,66, No. 1, 25–32 (1992).
V. E. Kuzmin, D. V. Naroditskii, and I. S. Rublev ——ibid.,68, No. 6, 1044–1048 (1994).
Additional information
A. V. Bogatskii Physicochemical Institute, Ukrainian Academy of Sciences. Translated fromZhurnal Strukturnoi Khimii, Vol. 36, No. 5, pp. 879–883, September–October, 1995.
Translated by L. Smolina
Rights and permissions
About this article
Cite this article
Kuzmin, V.E., Rublev, I.S., Naroditskii, D.V. et al. Geometrical design of ligands for spherical ions. J Struct Chem 36, 798–802 (1995). https://doi.org/10.1007/BF02579671
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02579671