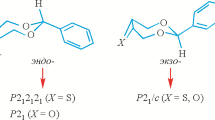
Abstract
This paper examines the possibility of classifying chiral simplices into homochiral subclasses. It is shown that chiral simplices can skip achiral intermediates to be transformed into another type of homochiral moleucles (stereochemical tunneling).
Similar content being viewed by others
References
V. I. Sokolov,Introduction to Theoretical Chemistry [in Russian], Nauka, Moscow (1973).
E. Ruch,Theor. Chim. Acta,11, No. 1, 183–189 (1968).
K. Mislow and P. Poggi-Corradini,J. Math. Chem.,13, No. 2, 209–211 (1993).
V. E. Kuzmin,Zh. Fiz. Khim.,68, No. 6 1037–1043 (1994).
V. E. Kuzmin, L. P. Trigub, I. V. Yudanova, et al.Chirality [in press].
IUPAC Chemical Nomenclature [in Russian], Vol. 3, VINITI Moscow (1983).
V. A. Nikanorov,Abstracts of Papers from the 5th All-Union Conference on Crystal Chemistry, Chernoglovka (1987), pp. 13, 191.
V. E., Kuzmin and I. B. Stelmakh,Zh. Strukt. Khim,28, 45 (1987).
V. E. Kuzmin, I. B. Stelmakh, D. V. Pozigun, and M. B. Bekker,J. Phys. Org. Chem.,5, 293 (1992).
D. I. Blokhinstev,Basic Quantum Mechanics [in Russian], Nauka, Moscow (1976).
Additional information
A. V. Bogatskii Physiochemical Institute, Ukrainian Academy of Sciences. Translated fromZhurnal Strukturnoi Khimii, Vol. 36, No. 5, pp. 873–878, September–October, 1995.
Translated by L. Smolina
Rights and permissions
About this article
Cite this article
Kuzmin, V.E. Homo- and heterochirality of dissymmetric tetrahedra (chiral simplices). Stereochemical tunneling. J Struct Chem 36, 793–797 (1995). https://doi.org/10.1007/BF02579670
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02579670