Summary
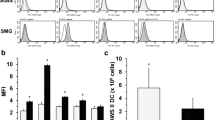
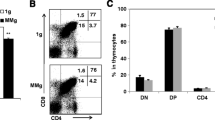
Generation of an effective immune response requires that antigens be processed and presented to T lymphocytes by antigen-presenting cells, the most efficient of which are dendritic cells (DC). Because of their influence on both the innate and the acquired arms of immunity, a defect in DC would be expected, to result in a broad impairment of immune function, not unlike that observed in astronauts during or after space flight. In the study reported here, we investigated whether DC generation and function are altered in a culture environment that models microgravity, i.e., the rotary-cell culture system (RCCS). We observed that RCCS supported the generation of DC identified by morphology, phenotype (HLA-DR+ and lacking lineage-associated markers), and function (high allostimulatory activity). However, the yield of DC from RCCS was significantly lower than that from static cultures. RCCS-generated DC were less able to phagocytoseAspergillus fumigatus conidia and expressed a lower density of surface HLA-DR. The proportion of Dc expressing CD80 was also significantly reduced in RCCS compared to static cultures. When exposed to fungal antigens, RCCS-generated DC produced lower levels of interleukin-12 and failed to upregulate some costimulatory/adhesion molecules involved in antigen presentation. These data suggest that DC generation, and some functions needed to mount an effective immune response to pathogens, may be disturbed in the microgravity environment of space.
Similar content being viewed by others
References
Banchereau, J.; Steinman, R. M. Dendritic cells and the control of immunity. Nature 392:245–252; 1998.
Cenci, E.; Mencacci, A.; Fè d'Ostiani, C., et al. Cytokine- and T helper-dependent lung mucosal immunity in mice with invasive pulmonary aspergillosis. J. Infect. Dis. 178:1750–1760; 1998.
Cooper, D.; Pellis, N. R. Suppressed PHA activation of T lymphocytes in simulated microgravity is restored by direct activation of protein kinase C. J. Leukoc. Biol. 63:550–562; 1998.
Davis, T. A.; Wiesmann, W.; Kidwell, W., et al. Effect of spaceflight on human stem cell hematopoiesis: suppression of erythropoiesis and myelopoiesis. J. Leukoc. Biol. 60:69–76; 1996.
Demotz, S.; Grey, H. M.; Sette, A. The minimal number of class II MHC-antigen complexes needed for T cell activation. Science 249:1028–1030; 1990.
Denning, D. W. Invasive aspergillosis. Clin. Infect. Dis. 26:781–803; 1998.
de Wynter, E. A.; Coutinho, L. H.; Pei, X., et al. Comparison of purity and enrichment of CD34+ cells from bone marrow, umbilical cord and peripheral blood (primed for apheresis) using five separation systems. Stem Cells 13:524–532; 1995.
Fernandez, N. C.; Lozier, A.; Flament, C., et al. Dendritic cells directly trigger NK cell functions: cross-talk relevant in innate anti-tumor immune responsesin vivo. Nat. Med. 5:405–411; 1999.
Grazziutti, M. L.; Rex, J. H.; Cowart, R. E., et al.Aspergillus fumigatus conidia induce a Thl-type cytokine response. J. Infect. Dis. 176:1579–1583; 1997.
Grouard, G.; Rissoan, M. C.; Filgueira, L., et al. The enigmatic plasmacytoid T cells develop into dendritic cells with interleukin (IL)-3 and CD40-ligand. J. Exp. Med. 185:1101–1111; 1997.
Hart, D. N. Dendritic cells: unique leukocyte populations which control the primary immune response. Blood 90:3245–3287; 1997.
Hershman, M. J.; Cheadle, W. G.; Wellhausen, S. R., et al. Monocyte HLADR antigen expression characterizes clinical outcome in the trauma patient. Br. J. Surg. 77:204–207; 1990.
Holt, P. G.; Schon-Hegrad, M. A.; McMenamin, P. G. Dendritic cells in the respiratory tract. Int. Rev. Immunol. 6:139–149; 1990.
Ichiki, A. T.; Gibson, L. A.; Jago, T. L., et al. Effects of spaceflight on rat peripheral blood leukocytes and bone marrow progenitor cells. J. Leukoc. Biol. 60:37–43; 1996.
Inaba, K.; Inaba, M.; Naito, M., et al. Dendritic cell progenitors phagocytose particulates, includingBacillus Calmette-Guerin organisms, and sensitize mice to mycobacterial antigens in vivo. J. Exp. Med. 178:479–488; 1993.
Kono, K.; Sekikawa, T.; Matsumoto, Y. Influence of surgical stress on monocytes and complications of infection in patients with esophageal cancer-monocyte HLA-DR antigen expression and respiratory burst capacity. J. Surg. Res. 58:275–280; 1995.
Konstantinova, I. V.; Rykova, M. P.; Lesnyak, A. T., et al. Immune changes during long-duration missions. J. Leukoc. Biol. 54:189–201; 1993.
Macatonia, S. E.; Hosken, N. A.; Litton, M., et al. Dendritic cells produce IL-12 and direct the development of Th1 cells from naive CD4+ T cells. J. Immunol. 154:5071–5079; 1995.
Meehan, R. T.; Neale, L. S.; Kraus, E. T., et al. Alteration in human mononuclear leukocytes following space flight. Immunology 76:491–497; 1992.
Nair, S. K.; Hull, S.; Coleman, D., et al. Induction of carcinoembryonic antigen (CEA)-specific cytotoxic T-lymphocyte responsesin vitro using autologous dendritic cells loaded with CEA peptide or CEA RNA in patients with metastatic malignancies expressing CEA. Int. J. Cancer 82:121–124; 1999.
Pellis, N. R.; Goodwin, T. J.; Risin, D., et al. Changes in gravity inhibit lymphocyte locomotion through type I collagen. In Vitro Cell. Dev. Biol. 33A:398–405; 1997.
Przepiorka, D.; van Vlasselaer, P.; Huynh, L., et al. Rapid debulking and CD34 enrichment of filgrastim-mobilized peripheral blood stem cells by semiautomated density gradient centrifugation in a closed system. J. Hematother. 5:497–502; 1996.
Rissoan, M. C.; Soumelis, V.; Kadowaki, N., et al. Reciprocal control of T helper cell and dendritic cell differentiation. Science 283:1183–1186; 1999.
Romani, N.; Gruner, S.; Brang, D., et al. Proliferating dendritic cell progenitors in human blood. J. Exp. Med. 180:83–93; 1994.
Sallusto, F.; Cella, M.; Danieli, C., et al. Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class II compartment: downregulation by cytokines and bacterial products. J. Exp. Med. 182:389–400; 1995.
Sallusto, F.; Lanzavecchia, A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J. Exp. Med. 179:1109–1118; 1994.
Sato, M.; Iwakabe, K.; Kimura, S., et al. Functional skewing of bone marrow-derived dendritic cells by Th1- or Th2-inducing cytokines. Immunol. Lett. 67:63–68; 1999.
Savary, C. A.; Grazziutti, M. L.; Melichar, B., et al. Multidimensional flowcytometric analysis of dendritic cells in peripheral blood of normal donors and cancer patients. Cancer Immunol. Immunother. 45:234–240; 1998.
Schwarz, R. P.; Goodwin, T. J.; Wolf, D. A. Cell culture for three-dimensional modeling in rotating-wall vessels: an application of simulated microgravity. J. Tissue Cult. Methods 14:51–58; 1992.
Siena, S.; Di Nicola, M.; Bregni, M., et al. Massive ex vivo generation of functional dendritic cells from mobilized CD34+ blood progenitors for anticancer therapy. Exp. Hematol. 23:1463–1471; 1995.
Sonnenfeld, G.; Davis, S.; Taylor, G. R., et al. Effect of space flight on cytokine production and other immunologic parameters of rhesus monkeys. J. Interferon Cytokine Res. 16:409–415; 1996.
Steinman, R. M. The dendritic cell system and its role in immunogenicity. Annu. Rev. Immunol. 9:271–296; 1991.
Szabo, M. C.; Teague, T. K.; McIntyre, B. W. Regulation of lymphocyte pseudopodia formation by triggering the integrin α1/β1. J. Immunol. 154:2112–2124; 1995.
Taylor, G. R.; Janney, R. P. In vivo testing confirms a blunting of the human cell-mediated immune mechanism during space flight. J. Leukoc. Biol. 51:129–132; 1992.
Taylor, G. R.; Konstantinova, I.; Sonnenfeld, G., et al. Changes in the immune system during and after spaceflight. Adv. Space Biol. Med. 6:1–32; 1997.
Wu, A. H. B.; Taylor, G. R.; Graham, G. A., et al. The clinical chemistry and immunology of long-duration space missions. Clin. Chem. 39:22–36; 1993.
Zhou, L. J.; Tedder, T. F. CD14+ blood monocytes can differentiate into functionally mature CD83+ dendritic cells. Proc. Natl. Acad. Sci. USA 93:2588–2592; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Savary, C.A., Grazziutti, M.L., Przepiorka, D. et al. Characteristics of human dendritic cells generated in a microgravity analog culture system. In Vitro Cell.Dev.Biol.-Animal 37, 216–222 (2001). https://doi.org/10.1007/BF02577532
Issue Date:
DOI: https://doi.org/10.1007/BF02577532