Abstract
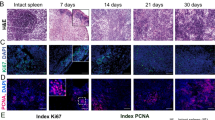
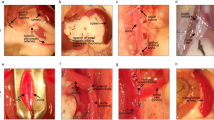
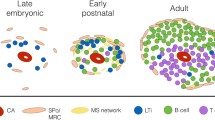
Splenic regeneration represents an interesting phenomenon both in relation to its role as a model system (to study the development of the complex three-dimensional architecture of an immunological organ) and because of the clinical application, namely autotransplantation of spleen. The latter is one of the attempts to restore splenic functions after splenectomy, which is known to increase a life-long risk of fatal sepsis. However, splenic functions of autotransplanted splenic tissue are known to be highly dependent on the recovery of the complex microenvironment and immunoarchitecture of the splenic compartments during the regeneration processes, but the elements inducing splenic reorganization are still unknown. Therefore, the present work investigates whether splenic stroma depleted of cells is able to induce regenerative processes after implantation. In addition, we tried to recombine stromal tissue with selected cell populations to study their influence. Cell-free stromal tissue induced angiogenesis and to a lesser extent also attracted the immigration of lymphocytes during the first 60 days of regeneration. However, after this period of regeneration, the transplants began to degenerate and were resorbed. The recombination of stromal tissue with mitogen-stimulated spleen cells only resulted in intensifying the degenerative processes, and all implants were resorbed after 120 days. Except that in the first 30 days there were some accumulations of lymphocytes that resembled primitive follicles, no splenic compartments such as red pulp, periarteriolar lymphoid sheath, or marginal zone could be detected in any of the transplants. From these results it can be concluded that splenic stroma can induce the primary events of splenic regeneration (like angiogenesis), but is not able to provide an appropriate microenvironment and immunoarchitecture for a correct repopulation and differentiation of cells. Furthermore, the recombination experiments point to a minor role of T-cells and possibly an important role for accessory cells in splenic regeneration.
Similar content being viewed by others
References
Bowdler A (1990) The spleen. Chapman & Hall New York
Barclay AN (1981) The localization of populations of lymphocytes defined by monoclonal antibodies in the rat lymphoid tissue. Immunology 42:593–600
Barclay AN (1981) Different reticular cells in rat lymphoid tissue identified by localization of Ia, Thy-1 and MRC OX-2 antigen. Immunology 44:727–736
Steinmann RM (1991) The dendritic cell system and its role in immunogenicity. Ann Rev Immunol 9:271–296
Dijkstra CD, Döpp EA (1982) Ontogenetic development of T- and B-lymphocytes and non-lymphoid cells in the white pulp of the rat spleen. Cell Tissue Res 222:69–79
Dijkstra CD, Döpp EA, Joling P, Kraal G (1985) The heterogeneity of mononuclear phagocytes in lymphoid organs: distinct macrophage subpopulations in the rat recognized by monoclonal antibodies ED1, ED2 and ED3. Immunology 54:589–599
Pellas TC, Weiss L (1990) Migration pathways of recirculating murine B cells and CD4+ and CD8+ lymphocytes. Am J Anat 187:355–373
Van den Eertwegh AJM, Claassen E (1991) T-cells in the spleen: localization, cytokine production and cell/cell interactions. Res Immunol 142:334–339
Di Cataldo A, Puelo S, Li Destri G, Racalbuto A, Trombatore G, Latteri F., Rodolico G (1987) Splenic trauma and overwhelming postplenectomy infection. Br J Surg 74: 343–345
Pimpl W, Dapunt O, Kaind H, Thalhamer J (1989) Incidence of septic and thromoembolic related deaths after splenectomy in adults. Br J Surg 76:517–521
King H, Shumaker HB (1952) Splenic studies: susceptibility to infection after splenectomy performed in infancy. Am Surg 136:239–242
Thalhamer J, Leitner W, Kurz ME, Liaunigg A, Seifriedsberger M, Bergmann ES, Kaindl H, Pimpl W (1992) Immunoarchitecture and specific functions of, splenic autotransplants at different implantation sites. Eur Surg Res 24:22–36
Liaunigg A, Kastberger C, Leitner W, Kurz M, Bergmann ES, Seifriedsberger M, Weinlich D, Pimpl W, Thalhamer J (1992), Regeneration of autotransplanted splenic tissue at different implantation sites. Cell Tissue Res 269:1–11
Thalhamer J, Lenglachner C, Grillenberger W, Pimpl W (1989) Alteration of proliferation and subtle changes of the protein synthesis in autologous transplanted spleens. Ann Surg 210:630–636
Pabst R, Westermann J, Rothkötter HJ (1991) Immunoarchitecture of regenerated splenic and lymph node transplants. Int Rev Cytol 128:215–259
Dijkstra CD, Döpp EA, Langevoort HL (1983) Regeneration of splenic tissue after autologous implantation: homing of T- and B- and Ia-positive cells in the white pulp of the rat spleen. Cell Tissue Res 229:97–107
Mishell BB, Shiigi SM (1980) Selected methods in cellular immunology. Freeman, San Francisco
Burck HC (1988) Histologische Technik. Thieme, Stuttgart
Weibel ER (1979) Practical methods for biological morphometry. In: Weibel ER (ed) Stereological methods, vol 1. Academic Press, New York, p 415
Sasaki K (1990) Splenic autotransplantation in nude mice: the formation of white pulp. APMIS 98:507–513
Willführ KU, Westermann J, Pabst R (1990) Absolute numbers of lymphocyte subsets migrating through the compartments of the normal and transplanted rat spleen. Eur J Immunol 20:903–911
Goldsmith HS, Griffith AL, Catsimpoolas N (1986) Increased vascular perfusion after administration of an omental lipid fraction. Surg Gynecol Obstet 162:579–585
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leitner, W., Bergmann, E.S. & Thalhamer, J. Regeneration of splenic stromal elements. Res. Exp. Med. 194, 221–230 (1994). https://doi.org/10.1007/BF02576383
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02576383