Abstract
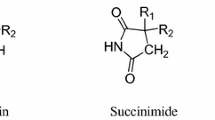
The X-ray structure of the title compound was performed to serve as the basis to find the “right” conformations among the minimum energy conformations derived from molecular modeling. The crystals of C21H16N2O2 are monoclinic, space groupP2l ln (Z=4);a=9.038(2),b=17.552(4),c=11.111(2)Å, β=91.36(3)°. The present study was performed on eight derivatives of diphenylsuccinimides, of which six were tested against the maximal electrical shock (MES). A theoretical test, based on the sign of the difference between the minima of the molecular electrostatic potential (MEP) at the carbonyl oxygens of the imide five-membered ring, designed for the purpose of an earlier study of phenylsuccinimides, was applied. This test confirmed the inactivity of the screened compounds and indicated that the remaining two diphenylsuccinimides are also inactive.
Similar content being viewed by others
References
Kwiatkowski, W. and Karolak-Wojciechowska, J.,SAR and QSAR in Envir. Res. 1993,1, 233.
Karolak-Wojciechowska, J.; Kwiatkowski, W.; Kieć-Kononowicz, K.Pharmazie 1995,50, 114.
Chmielewska, B.Pharmazie 1983,38, 872.
Obniska, J.; Zecj, A.; Karolak-Wojciechowska, J.Acta Polon. Pharm. Drug Research, in preparation.
Sheldrick, G.M.,Acta Crystallogr 1990,A46, 467.
Sheldrick, G.M.Program for the Refinement of Crystal Structures; Univ. of Göttingen: Germany, 1993.
Bukert, U.; Allinger, N.L.Molecular Mechanics. Am. Chem. Soc. Monograph 177, Washington, D.C., 1982.
PCMODEL. 4.Molecular modeling software for the IBM PC, Serena Software; Bloomington, 1990.
Steward, J.J.P.,Comput.-Aided. Mol. Des. 1990,4, 1.
MOPAC. 6. QCPE no.455.
Weinstein, H.; Osman, R.; Green J.P.; Topiel, S.,Chemical Applications of Atomic and Molecular Electrostatic Potentials; Plenum: New York, 1981; p 257.
Kwiatkowski, W. Ph.D. Thesis, Technical University: Łódź, 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Karolak-Wojciechowska, J., Błaszczyk, M., Kwiatkowski, W. et al. Electronic conditions for anticonvulsant activity of diphenylsuccinimides: Crystal and molecular structure of 1,1-diphenyl-N-(2-pyridyl)-succinimide. J Chem Crystallogr 27, 297–301 (1997). https://doi.org/10.1007/BF02575977
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02575977