Abstract
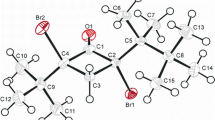
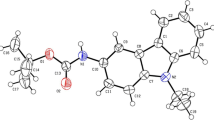
The cyclobutane ring displays a puckered conformation with the amide group in a perpendicular conformation. An intramolecular hydrogen bond held fixed the orientation of the 2-hydroxy-2-propyl moiety, causing the magnetic non equivalence of methyl groups observed in the room temperature1H-NMR spectrum. Crystal packing reveals the formation of bi-molecular layers parallel to thebc plane. The compound crystallizes in the space groupP21,/c witha=13.351(3),b=7.569(2),c=13.271(3)Å and β=92.03(3)o.
Similar content being viewed by others
References
Marvell, E.N.; Lin, C.J. Am. Chem. Soc. 1978,100, 877.
Maas, G.; Hummel, Ch.Chem. Ber. 1980,113, 3679.
Stork, G.; Cohen, J.F.J. Am. Chem. Soc. 1974,96, 5270.
Lallemand, J-Y.; Onaga, M.Tetrahedron Letts. 1975, 585.
Altomare, A.; Cascarano, G.; Giacovazzo, C.; Guagliardi, A.; Burla, M.C.; Polidori G.; Camalli, M.J. Appl. Cryst. 1994,27, 435.
Sheldrick, G.M.1990.SHELXTL/PC User's Manual; Siemens Analytical X-rays Instruments, Inc.: Madison WI.
Almenningen, A.; Bastiansen, O.; Skancke, P.N.Acta Chem. Scand 1961,15, 711.
Allen, F.H.Acta Crystallogr. 1984,B40, 64.
Allen, F.H.Acta Crystallogr. 1980,B36, 81.
Author information
Authors and Affiliations
Additional information
Contribution No. 1549 of Instituto de Química, UNAM.
Rights and permissions
About this article
Cite this article
Avila, G., Maldonado, L.A. & Toscano, R.A. 1-Phenyl-2t-(2-hydroxy-2-propyl)-1r-cyclobutanecarboxamide. J Chem Crystallogr 27, 125–128 (1997). https://doi.org/10.1007/BF02575905
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02575905