Summary
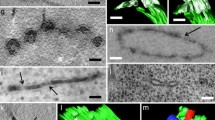
Transfer of membranes between endoplasmic reticulum and Golgi apparatus of the unicellular green alga,Micrasterias americana, is facilitated by 50–70 nm vesicles that form from part-rough. part-smooth transitional regions of the endoplasmic reticulum. In growing cells, the vesicles are present at the normal growth temperature of 23°C. However, at 16°C, vesicle accumulations occur. Golgi apparatus of non-growing cells exhibited both larger numbers of vesicles and larger dictyosomes at all temperatures. In non-growing cells, vesicle numbers also were increased at 16°C. The 16°C block was reconstituted in a cell-free system using Golgi apparatus-and endoplasmic reticulum-enriched fractions prepared from suspension cultures. When incubated in the presence of ATP and cytosol, transitional endoplasmic reticulum fragments ofMicrasterias responded by formation of membrane blebs and vesicles resembling those seen in situ. When prepared from cells metabolically labeled with [3H]leucine, the isolated transition elements supported the transfer of radioactivity of Golgi apparatus preparations immobilized on nitrocellulose strips. The transfer was time-and temperaturedependent and stimulated by ATP. The ATP-dependent component of transfer expressed at 23°C was reduced or absent at temperatures of 16°C or below. This suggested that membrane transfer mediated by transition vesicles was the same rate-limiting step in endoplasmic reticulum to Golgi apparatus membrane trafficking both in situ and in the cell-free system. Growth, as evidenced by a progressively alteredMicrasterias morphology, was slowed at low temperatures but showed no abrupt temperature transition as seen with the vesicular traffic between the endoplasmic reticulum and the Golgi appatus.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Balch WE, Keller DS (1986) ATP-coupled transport of vesciular stomatitis virus G protein: functional boundaries of secretory compartments. J Biol Chem 261: 14690–14696
—, Dunphy WG, Braell WA, Rothman JE (1984) Reconstitution of the transport of protein between successive compartments of Golgi measured by the coupled corporation of N-acetyl glucosamine. Cell 39: 405–416
Brand M, Jansen E, Ploegh HL (1985) Effect of reduced temperature on glycoprotein (Ig. HLA) processing and transport in lymphoid cells. Mol Immunol 22: 787–794
Copeland CS, Zimmer KP, Wagner KR, Healey GA, Mellman I, Helenius A (1988) Folding, trimerization and transport are sequential events in the biogenesis of influenza virus hemagglutinin. Cell 53: 197–209
Drawert H, Mix M (1962) Zur Frage von Struktur und Funktion des “Golgiapparates” in Pflanzenzellen. Sitzungsber Gesell Beförd Ges Naturwiss Marburg 83/94: 361–382
Dunphy WG, Rothman JE (1985) Compartment organization of the Golgi stack. Cell 42: 13–21
Fries E, Lindstrom I (1986) The effects of low temperatures on intracellular transport of newly synthesized albumin and haptoglobin in rat hepatocytes. Biochem J 237: 33–39
Griffiths G, Pfeiffer S, Simons K, Matlin K (1985) Exit of newly synthesized membrane proteins from the trans-cisterna of the Golgi complex to the plasma membrane. J Cell Biol 101: 949–964
Holmes KV, Doller EW, Sturman LS (1981) Tunicamycin resistant glycosylation of a coronavirus glycoprotein: determination of a novel type of viral glycoprotein. Virology 115: 334–344
Kiermayer O (1970) Elektronenmikroskopische Untersuchungen zum Problem der Cytomorphogenese vonMicrasterias denticulata Bréb. I. Allgemeiner Überblick. Protoplasma 69: 97–132
— (1981) Cytoplasmic basis of morphogenesis inMicrasterias In: Kiermayer O (ed) Cytomorphogenesis in plants. Springer, Wien New York, pp 147–190 [Alfert M et al (eds) Cell biology monographs, vol 8]
Kjellbom P, Larsson C (1984) Preparation and polypeptide composition of chlorophyll-free plasma membranes from leaves of light-grown spinach and barley. Physiol Plant 62: 501–509
Lagunoff D, Wan H (1974) Temperature dependence of mast cell histamine secretion. J Cell Biol 61: 809–811
Luft JM (1961) Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol 9: 409–414
Matlin KS, Simons K (1983) Reduced temperature prevents transfer of a membrane glycoprotein to the cell surface but does not prevent terminal glycosylation. Cell 34: 233–243
Meindl U (1990) Effects of temperature on cytomorphogenesis and ultrastructure ofMicrasterias denticulata Bréb. Protoplasma 157: 3–18
Morré DJ, Paulik M, Nowack D (1986) Transition vesicle formation in vitro. Protoplasma 132: 110–113
—, Minnifield N, Paulik M, (1989) Identification of the 16°C compartment of the endoplasmic reticulum in rat liver and cultured hamster kidney cells. Biol Cell 67: 51–60
—, Penel C, Morré DM, Sandelius AS, Moreau P, Andersson B (1991) Cell-free transfer and sorting of membrane lipids in spinach: donor and acceptor specificity. Protoplasma 160: 49–64
Noguchi T (1978) Transformation of the Golgi apparatus in the cell cycle, especially at the resting and earliest developmental stages of a green alga,Micrasterias americana. Protoplasma 95: 73–88
Nowack DD, Morré DM, Paulik M, Keenan TW, Morré DJ (1987) Intracellular membrane flow: reconstitution of transition vesicle formation and function in a cell-free system. Proc Natl Acad Sci USA 84: 6098–6102
Paulik M, Nowack DD, Morré DJ (1988) Isolation of a vescicular intermediate in the cell-free transfer of membrane from transitional elements of the endoplasmic reticulum to Golgi apparatus cisternae of rat liver. J Biol Chem 263: 17738–17748
Provasoli U, Pintner IJ (1959) Artificial media for freshwater algae; problems and suggestions. In: Tryon CA, Hartman RT (eds) The ecology of algae. Spec Publ no 2, Pymatuning Laboratory of Field Biology, University of Pittsburgh, Pittsburgh, pp 84–96
Roland JC, Lembi Ca, Morré DJ (1972) Phosphotungstic acidchromic acid as a selective electron-dense stain for plasma membrane of plant cells. Stain Technol 47:195–200
Rothman JE (1987) Protein sorting by selective retention in the endoplasmic reticulum and Golgi stack. Cell 50: 521–522
Saraste J, Kuismanen E (1984) Pre-and post-Golgi vacuoles operate in the transport of Semliki Forest virus membrane glycoproteins to the cell surface. Cell 38: 535–549
Saraste J, Palade GE, Farquhar MG (1986) Temperature sensitive steps in the transport of secretory proteins though the Golgi complex in exocrine pancreatic cells. Proc Natl Acad Sci USA 83: 6425–6429
Smith PK, Krohn RI, Hermason GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150: 76–85
Tartakoff AM (1986) Temperature and energy dependence of secretory protein transport in the exocrine pancreas. EMBO J 5: 1477–1482
Tooze J, Tooze SA, Warren G (1984) Replication of coronavirus MHV-A59 in sac(−) cells: determination of the first site of budding of progeny virions. Eur J Cell Biol 33: 291–293
Tooze SA, Tooze J, Warren G (1988) Site of addition of N-acetyl-galactosamine to the E1 glycoprotein of mouse hepatitis virus-A59. J Cell Biol 106: 1475–1487
Author information
Authors and Affiliations
Additional information
Dedicated to the memory of Professor O. Kiermayer
Rights and permissions
About this article
Cite this article
Noguchi, T., Morré, D.J. Vesicular membrane transfer between endoplasmic reticulum and golgi apparatus of a green alga,Micrasterias americana . Protoplasma 162, 128–139 (1991). https://doi.org/10.1007/BF02562556
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02562556