Summary
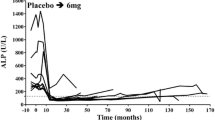
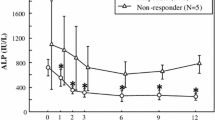
4-amino-1-hydroxybutylidene-1,1-bis-phosphonate (AHButBP) was given intravenously (2.5–25 mg/day for 4 days) to 14 patients with Paget's disease of bone, five of whom had been treated with dichloromethylidene bisphosphonate (Cl2MBP) 32 months earlier. In the nine patients who had not been treated previously with bisphosphonates, the short course of AHButBP induced a suppression of serum alkaline phosphatase and urinary hydroxyproline values down to 30% of initial values. The biochemical suppression of the disease was sustained for 2–18 months and the time to relapse did correlate to the logarithm of the dose (P<0.001). In the five patients previously treated for Paget's disease, an apparent resistence to treatment with AHButBP was observed. However, in these patients both serum alkaline phosphatase and urinary hydroxyproline fell to or even below the nadir values which had previously been achieved with Cl2MBP, irrespective of the degree of relapse. Thus the degree of suppression of Paget's disease of bone, achievable after treatment with bisphosphonates, seems to be constant for each patient, such that normal levels of serum alkaline phosphatase and urinary hydroxyproline cannot usually be attained in patients with extremely active disease.
Similar content being viewed by others
References
Douglas DL, Russell RGG, Preston CJ, Prenton MA, Duckworth T, Kanis JA, Preston FE, Woodhead SJ (1980) Effect of dichloromethylene disphosphonate in Paget's disease of bone and in hypercalcaemia due to primary hyperparathyroidism or malignant disease. Lancet 1:1043–1048
Frijlink WB, Bijvoet OLM, te Velde J, Heynen G (1979) Treatment of Paget's disease with (3-amino-1-hydroxypropylidene)-1,1-bisphosphonate (APD). Lancet 1:799–802
Johnston CC, Khairi MRA, Meunier PJ (1980) Use of etidronate (EHDP) in Paget's disease of bone. Arthritis Rheum 23:1172–1176
Jung A, Chantraine A, Donath A, van Ouwenaller C, Turnill D, Mermillod B, Kitler ME (1983) Use of dichloromethylene diphosphonate in metastatic bone disease. N Engl J Med 308:1499–1501
van Breukelen FMJ, Bijvoet OLM, van Oosterom AT (1979) Inhibition of osteolytic bone lesions by (3-amino-1-hydroxypropylidene)-1,1-bisphosphonate (APD). Lancet 1:803–805
Kanis JA (1984) Monitoring the treatment of Paget's disease with etidronate. Calcif Tissue Int 36:623–631
Bijvoet OLM, Frijlink WD, Jie K, Van Der Linden H, Meijer CJLM, Mulder H, Van Paassen HG, Reitsma PH, te Velde J, Devries E, Van Den Wey JP (1980) APD in Paget's disease of bone. Role of the mononuclear phagocyte system? Arthritis Rheum 23:1193–1204
Yates AJP, Percival RC, Gray RES, Atkins RM, Urwin GH, Hamdy NAT, Preston Cj, Beneton MNC, Russell RGG, Kanis JA (1985) Intravenous clodronate in the treatment and retreatment of Paget's disease of bone. Lancet 1:1474–1477
Reitsma PH, Teitelbaum SL, Bijvoet OLM, Kahn A (1982) Differential action of the bisphosphonates (3-amino-1-hydroxypropylidene)-1,1-bisphosphonate (APD) and disodium dichloromethylidene bisphosphonate (Cl2MDP) on rat macrophage-mediated bone resorption in vitro. J Clin Invest 70:927–933
Adami S, Guarrera G, Spiazzi G, Salvagno G, Marini G, Rosini S, Lo Cascio V (1984) Sequential treatment of Paget's disease with human calcitonin and dichloromethylene-disphosphonate. Metab Bone Dis Rel Res 5:131–137
Yakatan GJ, Poynor WJ, Talbert RL, Floyd BF, Slorgn CL, Ampuiski RS, Benedict JJ (1982) Clodronate kinetics and bioavailability. Clin Pharmacol Ther 31:402–410
Michael WR, King WR, Wakin JM. (1972) Metabolism of disodium ethane-1-hydroxy-1,1-diphosphonate (disodium etidronate) in the rat, rabbit, dog and monkey. Toxic Appl Pharmacol 21:503–515
Kanis JA, Evanson JM, Russell RGG (1981) Paget's disease of bone: diagnosis and management. Metab Bone Dis Rel Res 4 and 5:219–230
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Adami, S., Salvagno, G., Guarrera, G. et al. Treatment of Paget's disease of bone with intravenous 4-amino-1-hydroxybutylidene-1,1-bisphosphonate. Calcif Tissue Int 39, 226–229 (1986). https://doi.org/10.1007/BF02555208
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02555208