Abstract
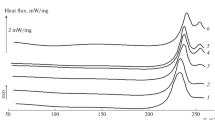
Water molecules in hydrogels were classified into three categories according to phase transition behavior; non-freezing, freezing bound and free water. Melting, crystallization, and glass transition of water in hydrogels reflected the state of the water interacting with polysaccharides. Freezing bound water formed metastable ice by slow cooling and formed amorphous ice by quenching. From the isothermal crystallization measurement, nucleation rate and crystal growth rate were obtained. The crystal growth rate of freezing bound water was about ten times slower than that of free water. The DSC characterization of water in hydrogels was summarized.
Zusammenfassung
In Übereinstimmung mit dem Phasenum wandlungsverhalten werden Wassermoleküle in Hydrogelen in drei Kategorien eingestuft; nicht gefrierendes, gefrierendes gebundenes und freies Wasser. Das Schmelzen, die Kristallisation und die Glasumwandlung von Wasser in Hydrogen wiederspiegeln den Zustand von Wasser, welches in Wechselwirkung mit Polysacchariden tritt. Gefrierendes gebundenes Wasser bildet metastabiles Eis durch langsames Abkühlen und amorphes Eis durch Abschrecken. Anhand isothermer Kristallisationsmessungen wurden Keimbildungsgeschwindigkeit und Kristallwachstumsgeschwindigkeit erhalten. Die Kristallwachstumsgeschwindigkeit von gefrierendem gebundenem Wasser liegt zehnmal niedriger als die von freiem Wasser.
Similar content being viewed by others
References
J. D. Andrade, ‘Hydrogels for Medical and Related Applications’, ACS Symp. Ser. 31, Washington DC: American Chemical Society, 1976.
H. Yoshida and Y. Miura, J. Memb. Sci., 68 (1992) 1.
K. Nishinari, J. Soc. Rheol. Jpn, 17 (1989) 100.
K. A. Mauritz and R. M. Fu, Macromolecules, 21 (1988) 1324.
W. B. Wise and P. E. Pfeffer, Macromolecules, 20 (1987) 1550.
F. X. Quinn, E. Kampff, G. Smith and M. McBrierty, Macromolecules, 21 (1988) 3192.
H. Uedaira, Hyomen, 13 (1975) 297.
K. Nakamura, T. Hatakeyama and H. Hatakeyama, Polymer, 22 (1981) 473.
K. Nakamura, T. Hatakeyama and H. Hatakeyama, Tex. Res. J., 51 (1981) 607.
T. Hatakeyama, K. Nakamura, H. Yoshida and H. Hatakeyama, Thermochim Acta, 88 (1985) 223.
T. Hatakeyama, H. Hatakeyama and H. Hatakeyama, Polymer, 28 (1987) 1282.
H. Yoshida, T. Hatakeyama and H. Hatakeyama, Polymer, 31 (1990) 693.
H. Yoshida, T. Hatakeyama and H. Hatakeyama, ACS Synposium Series 489 (1992) 217.
H. Yoshida, T. Hatakeyama and H. Hatakeyama, Koubunshi Ronbunsyu, 46 (1989) 597.
T. Hatakeyama, H. Yoshida and H. Hatakeyama ‘Physics and Chemistry of Ice’ Ed. by N. Maeno, Hokkaidou Univ. Press, Sapporo 1992, p. 262.
H. Yoshida, T. Hatakeyama and H. Hatakeyama, ‘Physics and Chemistry of Ice’, Ed. by N. Maeno, Hokkaidou Univ. Press, Sapporo 1992, p. 284.
S. Nakamura, M. Todoki, K. Nakamura and H. Kanetsuna, Thermochim Acta, 136 (1988) 363.
T. Hatakeyama and H. Kanetsuna, Thermochim Acta, 138 (1989) 327.
H. Yoshida, T. Hatakeyama and H. Hatakeyama, J. Intelligent Mat. Sci., to be published.
R. Kjellander and E. Florin, J. Chem. Soc., Faraday Trans, 77 (1981) 1.
H. Yoshida, T. Hatakeyama and H. Hatakeyama, Polymer Preprints Japan, 41 (1992).
L. Bosio, G. P. Johari, M. Oumezzine and J. Teixeira, Chem. Phys. Lett., 188 (1992) 113.
N. Okui, Polymer Bulletin, 23 (1990) 111.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshida, H., Hatakeyama, T. & Hatakeyama, H. Characterization of water in polysaccharide hydrogels by DSC. Journal of Thermal Analysis 40, 483–489 (1993). https://doi.org/10.1007/BF02546617
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02546617