Abstract
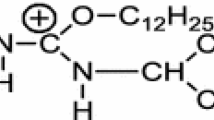
The properties in aqueous solution of the series of sodium sulfoalkyl alkanoates of general formula CnH2n+1COO(CH2)mSO3Na (where n=9,10, and 11; m=2,3, and 4) have been investigated by measurements of surface tension. According to the Gibbs absorption isotherm equation, the slope of the curves of the surface tension vs. logarithm concentration gives the amount of surfactant adsorbed. The surface tension of sodium 4-sulfobutyl ester derivatives was lower than that of sodium 2-sulfoethyl and 3-sulfopropyl ester derivatives in the region below the critical micelle concentration. The areas occupied/molecule for sodium 2-sulfoethyl and 3-sulfopropyl ester derivatives were in the range 51–56 Å2, which agreed with the value 50 Å2 obtained for the sodium dodecyl sulfate. However, the values of the areas occupied/molecule for sodium 4-sulfobutyl ester derivatives were 63–65 Å2, which was large compared with that of sodium dodecyl sulfate. The effect of the ester group in the sodium 4-sulfobutyl ester derivatives on the orientation of the molecule at the air-water interface is different from the effect of the ester group in the sodium 2-sulfoethyl and 3-sulfopropyl ester derivatives on the orientation of the molecule at the air-water interface.
Similar content being viewed by others
References
Hikota, T., and K. Meguro, JAOCS 47:197 (1970).
Hikota, T., Bull. Chem. Soc. Japan 43:2236 (1970).
Robins, D.C., and I.L. Thomas, J. Colloid and Interface Sci. 26:407 (1968).
Dreger, E.E., G.I. Kelm, G.D. Miles, L. Shedlovsky and J. Ross, Ind. Eng. Chem., 36:610 (1944).
Phillips, J. Trans. Faraday Soc. 51:561 (1955).
Author information
Authors and Affiliations
About this article
Cite this article
Hikota, T., Meguro, K. Studies of ester-containing surfactants: Effect of position of ester group on surface tension. J Am Oil Chem Soc 51, 1–3 (1974). https://doi.org/10.1007/BF02545203
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02545203