Summary
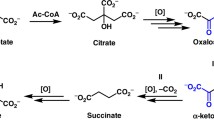
From the oxidation of malate and citrate (instead of isocitrate) by livingE. coli cells no formation of glyoxylate could be observed. On the other hand glyoxylate was always formed in the presence of acetate or glycolate. This result is further proof that glyoxylate is a direct intermediate of the oxidation of acetate and glycolate according to the monocarboxylic acid scheme.
Similar content being viewed by others
References
Ajl, S. J. 1958. Physiol. Reviews38, 196.
Bolcato, V., Boschetti, E. andMontoya, E. 1956a. Antonie van Leeuwenhoek22, 131.
Bolcato, V., Fraschini, M. andBoniperti, G. 1956b. Antonie van Leeuwenhoek22, 419.
Bolcato, V. 1959. Experientia, in press.
Bolcato, V., de Bernard, B. andLeggiero, G. 1957. Arch. Biochem. Biophys.69, 372.
Bolcato, V. andLeggiero, G. 1958. Annali di Chimica48, 177.
Kornberg, H. L. andMadsen, N. B. 1957. Biochim. Biophys. Acta24, 651.
Kornberg, H. L. andMadsen, N. B. 1958. Biochem. J.68, 549.
Utter, M. F. 1958. Ann. Review of Biochem.27, 274.
Wong, D. T. O. andAjl, S. J. 1957. Science126, 1013.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bolcato, V. Glyoxylate intermediate of the direct oxidation of acetate and glycolate byE. coli . Antonie van Leeuwenhoek 25, 179–182 (1959). https://doi.org/10.1007/BF02542844
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02542844