Abstract
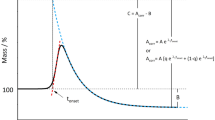
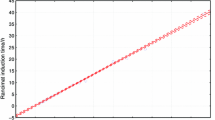
A method for evaluating the oxidative stability of oils based on heating the sample in an oven and periodically testing for weight gain has been known for over 100 years. Thermogravimetric analysis (TGA) with its highly sensitive recording electrobalance turns this simple method into a powerful technique for studying the relative thermooxidative stabilities of oils. This paper explores the potential of an isothermal TGA via evaluation of several freshly processed, unhydrogenated soybean oils. The objective was to define the parameters of the TGA experiment for fast routine measurement and to compare isothermal and dynamic experiments. The consideration of influence of temperature, specific surface area, air flow rate, sample volatilization and nature of the sample pan on the induction period (IP), which is a measure of the oil’s resistance to oxidation, revealed that the temperature and specific surface area are the major parameters that affect the oxidation process. The Arrhenius plot in the range of 80–150 C has shown that the overall activation energy of the oxidation process for three freshly processed oils lies within the range of 21–22 kcal/mole. The rate of oxidation at 150 C is directly proportional to the specific surface area of an oil, suggesting that the rate of oxygen diffusion determines reaction rates at this temperature. A coefficient of variation of the IP better than 2% was found in a routine experiment (10 mg sample, 150 C, 60 ml/min air flow rate, aluminum pan) for typical oils which had an IP range of 25–31 min.
Similar content being viewed by others
References
Gray, J.I., JAOCS 55:539 (1978).
Hadorn, H., and K. Zurcher, Dtsch. Lebensm. Rundschau 70:57 (1974).
Wheeler, D.H., Oil and Soap 9:89 (1932).
AOCS Official and Tentative Methods, Champaign, IL, 1981, Method Cd 12–57.
deMan, J.M., and L. deMan, JAOCS 61:534 (1984).
“Petroleum Products and Lubricants,” Part 24, Annual Book of Standards, American Society for Testing and Materials, ASTM, Philadelphia, PA, 1972, Method D 2272-67 (Reapproved 1983).
Gearhart, W.M., B.N. Stuckey and J.J. Austin, JAOCS 34:427 (1957).
Warner, K., and E.N. Frankel, JAOCS 62:100 (1985).
Cross, C.K., JAOCS 47:229 (1970).
Hassel, R.L., JAOCS 53:179 (1976).
Nieschlag, H.J., J.W. Hagemann and J.A. Rothfus, Anal. Chem. 46:2215 (1974).
Hagemann, J.W., and J.A. Rothfus, JAOCS 56:629 (1979).
Buzás, I., J. Simon and J. Holló, J. Thermal. Anal. 12:397 (1977).
Buzás, I., E. Kurucz-Lusztig and J. Holló, Acta Aliment. Acad. Sci. Hung. 7:335 (1978).
Buzás, I., E. Kurucz and J. Holló, JAOCS 56:685 (1979).
Olcott, H.S., and E. Einset, JAOCS 35:161 (1958).
Cassel, B.B., and A.P. Gray, Thermochim. Acta 36:265 (1980).
Norem, S.D., M.J. O’Neill and A.P. Gray, Thermochim. Acta 1:29 (1970).
Kwon, T.W., H.E. Snyder and H.G. Brown, JAOCS 61:1843 (1984).
Going, L.H., JAOCS 45:632 (1968).
Rock, S.P., and H. Roth, JAOCS 41:228 (1964).
Emanuel, N.M., “The Inhibition of Fat Oxidation Processes,” Pergamon Press, Oxford, New York, 1967, pp. 29–33.
Author information
Authors and Affiliations
About this article
Cite this article
Mikula, M., Khayat, A. Reaction conditions for measuring oxidative stability of oils by thermogravimetric analysis. J Am Oil Chem Soc 62, 1694–1698 (1985). https://doi.org/10.1007/BF02541668
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02541668