Abstract
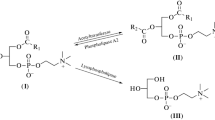
A series of halogen-containing alkylglycerolipid analogs has been checked for their cytostatic activity both in vitro and in vivo. The compounds included alkyldeoxyhaloglycerols (I), alkyldeoxyhaloglycerophosphocholines (II), and alkyldeoxyhaloglycerophosphoric acids and alkyl esters (III). While compounds I and III were moderately active, compounds II were found to have a strong inhibitory effect on the proliferation of Ehrlich ascites tumor cells in vitro. Cell growth inhibition of 50% or more was found mainly in the late S- or G2-phase of the cell cycle as revealed by flow cytometry. Alkyl lysophospholipid analogs II and cholesterol form liposomes with high encapsulation efficiency and low permeability for entrapped substances. Compounds II were active against Lewis lung carcinoma in mice when applied in free form or as liposomes.
Similar content being viewed by others
Abbreviations
- ALP:
-
alkyl lysophospholipids
- CF:
-
6-carboxyfluorescein
- CH:
-
cholesterol
- DG:
-
diacylglycerol
- EAT cells:
-
Ehrlich ascites tumor cells
- EPC:
-
egg phosphatidylcholine
- HEPC:
-
hydrogenated egg phosphatidylcholine; ip, intraperitoneal(ly)
- IP3 :
-
inositol-1,4,5-triphosphate
- MLV:
-
multilamellar vesicles
- PA:
-
phosphatidic acid
- PI:
-
phosphatidylinositol
- PIP:
-
phosphatidylinositol-4-phosphate
- PIP2 :
-
phosphatidylinositol-4,5-diphosphate
- PKC:
-
proteinkinase C
- SUV:
-
small unilamellar vesicles
References
Langen, P. (1975)Antimetabolites of Nucleic Acid Metabolism, Gordon and Bresch, New York, London and Paris
Ferber, E., Körner, E., Schmidt, B., Peskar, B.A., and Anders, C. (1980) inMembrane Fluidity: Biophysical Techniques and Cellular Regulation, Part IV: Phospholipid Changes Accompanying Physiological Events, (Kates, M., and Kuksis, A., eds.) pp. 239–263, The Human Press, Inc., Clifton, NJ.
Nishizuka, Y. (1984)Science 225, 1365–1370.
Munder, P.G., Modolell, M., Bausert, W., Oettgen, H.F., and Westphal, O. (1981) inAugmenting Agents in Cancer Therapy, (Hersh, E.M., ed.) pp. 441–467, Raven Press, New York.
Berdel, W.E. (1982)Blutt 44, 71–78.
Kraft, R., Brachwitz, H., Etzold, G., Langen, P., and Zöpfl, H.J. (1979)J. Prakt. Chem. 321, 756–768.
Brachwitz, H., Kraft, R., Langen, P., Etzold, G., and Schildt, J. (1979)J. Prakt, Chem. 321, 769–774.
Brachwitz, H., Langen, P., Otto, A., and Schildt, J. (1979)J. Prakt. Chem. 321, 775–786.
Brachwitz, H., Langen, P., Hintsche, R., and Schildt, J. (1982)Chem. Phys. Lipids 31, 33–52.
Brachwitz, H., Langen, P., and Schildt, J. (1984)Chem. Phys. Lipids 34, 355–362.
Brachwitz, H., Franke, P., Hintsche, R., Langen, P., and Schildt, J. (1984)Chem. Phys. Lipids 36, 137–152.
Langen, P., Brachwitz, H., and Schildt, J. (1979)Acta Biol. Med. Germ. 38, 965–974.
Müller, E., Hegewald, H., Jaroszewicz, K., Kumme, G., Hoppe, H., and Frunder, H. (1986)Biochem. J. 235, 775–783.
Uchida, T., and Filburn, C.R. (1984)J. Biol. Chem. 259, 12311–12314.
Nuhn, P., Schenk, P., Richter, H., Arndt, D., Jandrig, J., and Winsel, K. (1985)Pharmazie 40, 705–709.
Soodsma, J.F., Piantadosi, C., and Snyder, F. (1970)Cancer Res. 30, 309–311.
Modolell, M., Andreesen, R., Pahlke, W., Brugger, U., and Munder, P.G. (1979)Cancer Res. 39, 4681–4686.
Helfman, D.M., Barnes, K.C., Kindade, J.M., Vogler, W.R., Shoji, M., and Kuo, J.F. (1983)Cancer Res. 43, 2955–2961.
Honma, Y., Kasukabe, T., Hozumi, M., Tsushima, S., and Nomura, H. (1981)Cancer Res. 41, 3211–3216.
Langen, P., Brachwitz, H., Schildt, J., Waschke, S., and Guenther, W. (1981) DDR-Patent 0153116.
Ostermann, G., Brachwitz, H., and Till, U. (1984)Biomed. Biochim. Acta 43, 349–355.
de Chaffey de Courcelles, D., Raevens, P., and Van Belle, H. (1984)Biochem. Biophys. Res. Commun. 123, 589–594.
Berger, M.R., and Schmähl, D. (1986)J. Cancer Res. Clin. Oncol. 111 Suppl., 47.
Author information
Authors and Affiliations
About this article
Cite this article
Brachwitz, H., Langen, P., Arndt, D. et al. Cytostatic activity of syntheticO-alkylglycerolipids. Lipids 22, 897–903 (1987). https://doi.org/10.1007/BF02535551
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02535551