Abstract
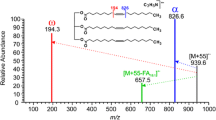
Synthetic mixtures of C40 to C47 sterol esters in groups of 7 esters were effectively separated and analyzed by capillary gas chromatography-mass spectrometry. Ammonia chemical ionization of all 20 sterol esters analyzed at a source block temperature of 120 C yielded (M+NH4)+ and (M+H-RCO2H)+ ions of high abundance or as base peak, thereby indirectly indicating the molecular weights of the ester and the sterol and acid moieties. Ammonia CI spectra of all esters containing a Δ5-sterol moiety exhibited in addition to the above 2 ions an M+NH4-RCO2 H fragment. At a source block temperature of 150 C, M+H-RCO2 H fragment was the base peak for all esters, and there was little or no indication of an (M+NH4)+ adduct ion. Protonated molecules were not observed for any esters analyzed by methane or isobutane CI. Molecular ions of 3–14% intensity were obtained for only 3 of the esters analyzed by electron impact; they contained a Δ7-bond in the sterol nucleus, and the acid moiety was either saturated normal or branched chain or contained a single double bond. The base peak was a function of both the acid and sterol moieties of the sterol ester. The esters containing both saturated straight chain acid and saturated sterol moieties exhibited a base peak at m/z 215. The Δ5-sterol esters with saturated branched or straight chain acid moieties exhibited base peaks at M-RCO2 H. Other ions also were of diagnostic value.
Similar content being viewed by others
References
Murata, T., Takahashi, S., and Takeda, T. (1975) Anal. Chem. 47, 577–580.
Smith, N.B. (1983) J. Chrom. 254, 195–202.
Wakeham, S.G., and Frew, N.M. (1982) Lipids 17, 831–843.
Smith, N.B. (1982) Lipids 17, 464–468.
Smith, L.L., Teng, J.I., Lin, Y.Y., Seitz, P.K., and McGehee, M.F. (1981) J. Steroid Biochem. 14, 889–900.
Thompson, R.H., Patterson, G., Thompson, M.J., and Slover, H.T. (1981) Lipids 16, 694–699.
Steele, J.A., and Mosettig, E. (1962) J. Org. Chem. 28, 571–572.
Allen, C.F., and Kalm, M.J. (1963) in Organic Syntheses collective volume IV (Rabjohn, N., ed.), pp. 618–619, John Wiley & Sons, Inc., New York.
Budzikiewicz, H., Djerassi, C., and Williams, D.H. (1964) Structural Elucidation of Natural Products by Mass Spectrometry, Holden-Day, Inc., San Francisco.
Heller, S.R., and Milne, G.W.A. (1978) EPA/NIH Mass Spectral Data Base, U.S. Government Printing Office, Washington, D.C.
Zaretskii, Z.V. (1976) Mass Spectrometry of Steroids, John Wiley & Sons, New York.
Knights, B.A. (1967) J. Gas Chrom., 273–282.
Hirano, Y., Djerassi, C., and Tecon, P. (1982) Abstracts 30th Ann. Confer. on Mass Spectrom. and Allied Topics, 143–144.
Tecon, P., Hirano, Y., and Djerassi, C. (1982) Org. Mass Spectrom. 17, 277–285.
Field, F.H. (1968) J. Am. Chem. Soc. 90, 5649–5656.
McKibben, G.H., Thompson, M.J., Parrott, W.L.; Thompson, A.C., and Lusby, W.R., manuscript in preparation.
Author information
Authors and Affiliations
About this article
Cite this article
Lusby, W.R., Thompson, M.J. & Kochansky, J. Analysis of sterol esters by capillary gas chromatography-electron impact and chemical lonization-mass spectrometry. Lipids 19, 888–901 (1984). https://doi.org/10.1007/BF02534520
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02534520